The borderline range of toxicological methods: Quantification and implications for evaluating precision
Main Article Content
Abstract
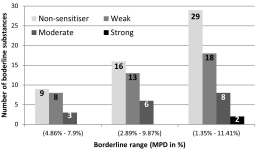
Test methods to assess the skin sensitization potential of a substance usually use threshold criteria to dichotomize continuous experimental read-outs into yes/no conclusions. The threshold criteria are prescribed in the respective OECD test guidelines and the conclusion is used for regulatory hazard assessment, i.e., classification and labelling of the substance. We can identify a borderline range (BR) around the classification threshold within which test results are inconclusive due to a test method’s biological and technical variability. We quantified BRs in the prediction models of the non-animal test methods DPRA, LuSens and h-CLAT, and of the animal test LLNA, respectively. Depending on the size of the BR, we found that between 6% and 28% of the substances in the sets tested with these methods were considered borderline. When the results of individual non-animal test methods were combined into integrated testing strategies (ITS), borderline test results of individual tests also affected the overall assessment of the skin sensitization potential of the testing strategy. This was analyzed for the 2-out-of-3 ITS: Four out of 40 substances (10%) were considered borderline. Based on our findings we propose expanding the standard binary classification of substances into “positive”/“negative” or “hazardous”/“non-hazardous” by adding a “borderline” or “inconclusive” alert for cases where test results fall within the borderline range.
Article Details
Articles are distributed under the terms of the Creative Commons Attribution 4.0 International license (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution and reproduction in any medium, provided the original work is appropriately cited (CC-BY). Copyright on any article in ALTEX is retained by the author(s).