The use of categorical regression in the assessment of the risks of nutrient deficiency and excess
Main Article Content
Abstract
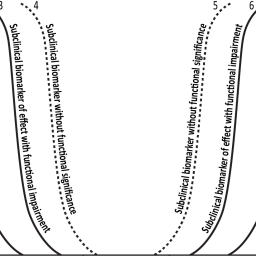
Exposure-response assessment methods have shifted towards more quantitative approaches, with health risk assessors exploring more statistically driven techniques. These assessments, however, usually rely on one critical health effect from a single key study. Categorical regression addresses this limitation by incorporating data from all relevant studies – including human, animal, and mechanistic studies – thereby including a broad spectrum of health endpoints and exposure levels for exposure-response analysis in an objective manner. Categorical regression requires the establishment of ordered response categories corresponding to increasingly severe adverse health outcomes and the availability of a comprehensive database that summarizes all data on different outcomes from different studies, including the exposure or dose at which these outcomes are observed and their severity. It has found application in the risk assessment of essential nutrients and trace metals. Since adverse effects may arise from either deficient or excess exposure, the exposure-response curve is U-shaped, which provides a basis for determining optimal intake levels that minimize the joint risks of deficiency and excess. This article provides an overview of the use of categorical regression fit exposure-response models incorporating data from multiple evidence streams. An extension of categorical regression that permits the simultaneous analysis of excess and deficiency toxicity data is presented and applied to comprehensive databases on copper and manganese. Future applications of categorical regression will be able to make greater use of diverse data sets developed using new approach methodologies, which can be expected to provide valuable information on toxic responses of varying severity.
Article Details

This work is licensed under a Creative Commons Attribution 4.0 International License.
Articles are distributed under the terms of the Creative Commons Attribution 4.0 International license (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution and reproduction in any medium, provided the original work is appropriately cited (CC-BY). Copyright on any article in ALTEX is retained by the author(s).
Academy of Medical Sciences (2007). Identifying the environmental causes of disease: How should we decide what to believe and when to take action? https://acmedsci.ac.uk/file-download/34590-IECDWebD.pdf
Aggett, P. J. (2007). Nutrient risk assessment: Setting upper levels and an opportunity for harmonization. Food Nutr Bull 28, Suppl 1, S27-S37. doi:10.1177/15648265070281s104
Allen, B., Zeiger, E., Lawrence, G. et al. (2005). Dose-response modeling of in vivo genotoxicity data for use in risk assessment: Some approaches illustrated by an analysis of acrylamide. Regul Toxicol Pharmacol 41, 6-27. doi:10.1016/j.yrtph.2004.09.006
Andersen, M. E., McMullen, P. D., Phillips, M. B. et al. (2019). Developing context appropriate toxicity testing approaches using new alternative methods (NAMs). ALTEX 36, 523-534. doi:10.14573/altex.1906261
ATSDR – Agency for Toxic Substances and Disease Registry (2000). Toxicological Profile for Manganese (Update). U.S. Department of Health and Human Services, Atlanta (GA). doi:10.15620/cdc:95222
ATSDR (2012). Toxicological Profile for Manganese. U.S. Department of Health and Human Services, Atlanta (GA).
Barnes, D. and Dourson, M. (1988). Reference dose (RfD): Description and use in health risk assessments. Regul Toxicol Pharmacol 8, 471-486. doi:10.1016/0273-2300(88)90047-5
Brown, K. G. and Strickland, J. A. (2003). Utilising data from multiple studies (meta-analysis) to determine effective dose-duration levels: Example: Rats and mice exposed to hydrogen sulfide. Regul Toxicol Pharmacol 37, 305-317. doi:10.1016/S0273-2300(03)00007-2
Brown, K. G. and Foureman, G. L. (2005). Concentration-time-response modelling for acute and short-term exposures. Regul Toxicol Pharmacol 43, 45-54. doi:10.1016/j.yrtph.2005.06.002
Chambers, A., Krewski, D., Birkett, N. et al. (2010). An exposure-response curve for copper excess and deficiency. J Toxicol Environ Health B Crit Rev 13, 546-578. doi:10.1080/10937404.2010.538657
Chifman J. and Laubenbacher, R. (2014). A systems biology approach to iron metabolism. Adv Exp Med Biol 844, 201-225. doi:10. 1007/978-1-4939-2095-2_10
Crump, K. S. (1984). A new method for determining allowable daily intake. Fundam Appl Toxicol 4, 854-871. doi:10.1016/0272-0590(84)90107-6
Davies, K. J. A. (2016). Adaptive homeostasis. Mol Aspects Med 49, 1-7. doi:10.1016/j.mam.2016.04.007
Deveau, M., Chen, C. P., Johanson, G. et al. (2015). The global landscape of occupational exposure limits-implementation of harmonization principles to guide limit selection. J Occup Environ Hyg 12, Suppl 1, S127-S144. doi:10.1080/15459624.2015.1060327
Edwards, L. M. (2017). Metabolic systems biology: A brief primer. J Physiol 595, 2849-2855. doi:10.1113/JP272275
EFSA NDA Panel – EFSA Panel on Nutrition, Novel Foods, and Food Allergens (2019). Scientific opinion on the dietary reference values for sodium. EFSA J 17, e05778. doi:10.2903/j.efsa.2019.5778
EFSA NDA Panel (2022). Guidance for establishing and applying tolerable upper intake levels for vitamins and essential minerals. EFSA J 20, e200102. doi:10.2903/j.efsa.2022.e200102
Gift, J. S., McGaughty, R., Singh, D. V. et al. (2008). Health assessment of phosgene: Approaches for the derivation of reference concentration. Regul Toxicol Pharmacol 51, 98-107. doi:10.1016/j.yrtph.2008.03.004
Haber, L., Strickland, J. A. and Guth, D. J. (2001). Categorical regression analysis of toxicity data. Comments Toxicol 7, 437-452.
Health Canada (1994). Risk assessment for the combustion products of methylcyclopentadienyl manganese tricarbonyl (MMT) in gasoline.
Hertzberg, R. C. and Miller, M. A. (1985). A statistical model for species extrapolation using categorical response data. Toxicol Ind Health 1, 43-57. doi:10.1177/074823378500100405
King, J. C. and Garza, C. (eds) (2007). International harmonisation of approaches for developing nutrient-based dietary standards. Food Nutr Bull 28, Suppl 1, S1-S151. doi:10.1177/15648265070281S101
Krewski, D., Chambers, A., Ransom Stern, B. et al. (2010). Development of a copper database for exposure-response analysis. J Toxicol Environ Health A 73, 208-216. doi:10.1080/15287390903340815
Krewski, D., Andersen, M. E., Tyshenko, M. G. et al. (2020). Toxicity testing in the 21st century: Progress in the past decade and future perspectives. Arch Toxicol 94, 1-58. doi:10.1007/s00204-019-02613-4
Krewski, D., Saunders-Hastings, P., Baan, R. A. et al. (2022). Development of an Evidence-Based Risk Assessment Framework. ALTEX 39, 667-693. doi:10.14573/altex.2004071
Lewis, J. and Dwyer, J. T. (2020). Establishing nutrient intake values. In B. P. Marriott, D. F. Birt, V. A. Stallings et al. (eds), Present Knowledge in Nutrition (267-287). Volume 2. London, UK: Elsevier. doi:10.1016/B978-0-12-818460-8.01001-7
Magistrato, A., Pavlin, M., Qasem, Z. et al. (2019). Copper trafficking in eukaryotic systems: Current knowledge from experimental and rotational efforts. Curr Opin Struct Biol 58, 26-33. doi:10. 1016/j.sbi. 2019.05.002
Mattison, D., Milton, B., Krewski, D. et al. (2016). Severity scoring of manganese health effects for categorical regression. Neurotoxicology 58, 203-216. doi:10.1016/j.neuro.2016.09.001
Mertz, W. (1993). Essential trace metals: New definitions based on new paradigms. Nutr Rev 51, 287-295. doi:10.1111/j.1753-4887.1993.tb03057.x
Milton, B., Farrell, P. J., Birkett, N. et al. (2017a). Modeling U-shaped exposure-response relationships for agents that demonstrate toxicity due to both excess and deficiency. Risk Anal 37, 265-279. doi:10.1111/risa.12603
Milton, B., Krewski, D., Mattison, D. R. et al. (2017b). Modelling U-shaped dose-response curves for manganese using categorical regression. Neurotoxicology 58, 217-225. doi:10.1016/j.neuro.2016.10.001
NRC – National Research Council (2000). DRI Dietary Reference Intakes: Applications in Dietary Assessment. National Academies Press, Washington, D.C.
NRC (2001). Dietary Reference Intakes for Vitamin A, Vitamin K, Arsenic, Boron, Chromium, Copper, Iodine, Iron, Manganese, Molybdenum, Nickel, Silicon, Vanadium, and Zinc. National Academies Press, Washington, D.C. doi:10.17226/10026
NRC (2017). Guiding Principles for Developing Dietary Reference Intakes Based on Chronic Disease. National Academies Press, Washington, D.C. doi:10.17226/24828
Ramoju, S. P., Mattison, D. R., Milton, B. et al. (2017). The application of PBPK models in estimating human brain tissue manganese concentrations. Neurotoxicology 58, 226-237. doi:10.1016/j.neuro.2016.12.001
Renwick, A. G., Flynn, A., Fletcher, R. J. et al. (2004). Risk-benefit analysis of micronutrients. Food Chem Toxicol 42, 1903-1922. doi:10.1016/j.fct.2004.07.013
Roels, H. A., Ghyselen, P., Buchet, J. P. et al. (1992). Assessment of the permissible exposure level to manganese in workers exposed to manganese dioxide dust. Br J Ind Med 49, 25-34. doi:10.1136/oem.49.1.25
Sand, S., Poirier, C. J. and Krewski, D. (2011). A signal-to-noise crossover dose as the point of departure for health risk assessment. Environ Health Perspect 119, 1766-1774. doi:10.1289/ehp.1003327
Sand, S., Parham, F. C. J., Portier, C. J. et al. (2017). Comparison of points of departure for health risk assessment based on high-throughput screening data. Environ Health Perspect 125, 623-633. doi:10.1289/ehp408
Schroeter, J. D., Nong, A., Yoon, M. et al. (2011). Analysis of manganese tracker kinetics and target tissue dosimetry in monkeys and humans with multi-route physiologically-based pharmacokinetic models. Toxicol Sci 120, 481-498. doi:10.1093/toxsci/kfq389
Smeraldi, C., Giarola, A., Aggett, P. J. et al. (2020). Use of mechanistic information to derive chemical specific adjustment factors – Refinement of risk assessment. Regul Toxicol Pharmacol 117, 104776. doi:10.1016/j.yrtph.2020.104776
Strickland, J. A. and Foureman, G. A. (2002). US EPA’s acute reference exposure methodology for acute inhalation exposures. Sci Total Environ 288, 51-63. doi:10.1016/S0048-9697(01)01114-7
Tyshenko, M. G., Willhite, D. Levy, L. et al. (2022). Evolution of the use of toxicological data in evaluating chemical safety. In W. Farland and D. Paustenback (eds.), Patty’s Toxicology. 7th edition. Wiley, New York, USA.
US EPA – U.S. Environmental Protection Agency (1993). Integrated Risk Information System (IRIS). 1.B. Reference concentration for chronic inhalation exposure (RfC). US EPA (U.S. Environmental Protection Agency) Substance Name: Manganese (CASRN – 7439-96-5).
US EPA – U.S. Environmental Protection Agency (1994). Re-evaluation of inhalation health risk association with methylcyclopentadienyl manganese tricarbonyl (MMT) in gasoline. Office of Research and Development, Environmental Criteria and Assessment Office, Research Triangle Park, NC (EPA 600/R-94/062).
WHO – World Health Organization (1996). Trace elements in human nutrition and human health. Geneva: World Health Organization.
WHO (2000). Manganese, Air Quality Guidelines for Europe. 2nd edition WHO Regional Office for Europe. Copenhagen, Denmark European Series No. 91, 154-156. ISBN 92 890 1358 3.
WHO/IPCS – WHO/International Program on Chemical Safety (2002). Principles and methods for the assessment of risk from essential trace elements (No. 228), World Health Organization. https://apps.who.int/iris/handle/10665/42416WHO
WHO (2006). A model for establishing upper levels of intake for nutrients and related substances. Report of a Joint FAO/WHO Technical Workshop on Nutrient Risk Assessment, World Health Organization.
Yetley, E. A., MacFarlane, A. J. Greene-Finestone, L. S. et al. (2017). Options for basing dietary reference intakes (DRIs) on chronic disease endpoints: Report from a joint US/Canadian-sponsored working group. Am J Clin Nutr 105, 249S-285S. doi:10.3945/ajcn.116.139097