Beyond pharmaceuticals: Fit-for-purpose new approach methodologies for environmental cardiotoxicity testing
Main Article Content
Abstract
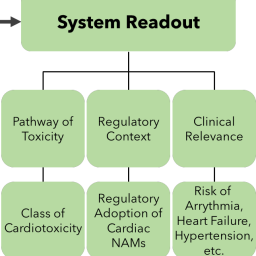
Environmental factors play a substantial role in determining cardiovascular health, but data informing the risks presented by environmental toxicants is insufficient. In vitro new approach methodologies (NAMs) offer a promising approach with which to address the limitations of traditional in vivo and in vitro assays for assessing cardiotoxicity. Driven largely by the needs of pharmaceutical toxicity testing, considerable progress in developing NAMs for cardiotoxicity analysis has already been made. As the scientific and regulatory interest in NAMs for environmental chemicals continues to grow, a thorough understanding of the unique features of environmental cardiotoxicants and their associated cardiotoxicities is needed. Here, we review the key characteristics of as well as important regulatory and biological considerations for fit-for-purpose NAMs for environmental cardiotoxicity. By emphasizing the challenges and opportunities presented by NAMs for environmental cardiotoxicity we hope to accelerate their development, acceptance, and application.
Article Details

This work is licensed under a Creative Commons Attribution 4.0 International License.
Articles are distributed under the terms of the Creative Commons Attribution 4.0 International license (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution and reproduction in any medium, provided the original work is appropriately cited (CC-BY). Copyright on any article in ALTEX is retained by the author(s).
Agarwal, A., Goss, J. A., Cho, A. et al. (2013). Microfluidic heart on a chip for higher throughput pharmacological studies. Lab Chip 13, 3599-3608. doi:10.1039/c3lc50350j
Ahmed, R. E., Anzai, T., Chanthra, N. et al. (2020). A brief review of current maturation methods for human induced pluripotent stem cells-derived cardiomyocytes. Front Cell Dev Biol 8, 178. doi:10.3389/fcell.2020.00178
Åkesson, A., Donat-Vargas, C., Berglund, M. et al. (2019). Dietary exposure to polychlorinated biphenyls and risk of heart failure – A population-based prospective cohort study. Environ Int 126, 1-6. doi:10.1016/j.envint.2019.01.069
Akins Jr, R. E., Rockwood, D., Robinson, K. G. et al. (2010). Three-dimensional culture alters primary cardiac cell phenotype. Tissue Eng Part A 16, 629-641. doi:10.1089/ten.tea.2009.0458
Alhamdow, A., Lindh, C., Albin, M. et al. (2017). Early markers of cardiovascular disease are associated with occupational exposure to polycyclic aromatic hydrocarbons. Sci Rep 7, 9426. doi:10.1038/s41598-017-09956-x
Alissa, E. M. and Ferns, G. A. (2011). Heavy metal poisoning and cardiovascular disease. J Toxicol 2011, 870125. doi:10.1155/2011/870125
Ando, H., Yoshinaga, T., Yamamoto, W. et al. (2017). A new paradigm for drug-induced torsadogenic risk assessment using human iPS cell-derived cardiomyocytes. J Pharmacol Toxicol Methods 84, 111-127. doi:10.1016/j.vascn.2016.12.003
Ankley, G. T., Bennett, R. S., Erickson, R. J. et al. (2010). Adverse outcome pathways: A conceptual framework to support ecotoxicology research and risk assessment. Environ Toxicol Chem 29, 730-741. doi:10.1002/etc.34
Archer, C. R., Sargeant, R., Basak, J. et al. (2018). Characterization and validation of a human 3d cardiac microtissue for the assessment of changes in cardiac pathology. Sci Rep 8, 1-15. doi:10.1038/s41598-018-28393-y
Bajaj, P., Schweller, R. M., Khademhosseini, A. et al. (2014). 3D biofabrication strategies for tissue engineering and regenerative medicine. Annu Rev Biomed Eng 16, 247-276. doi:10.1146/annurev-bioeng-071813-105155
Baker, B. M. and Chen, C. S. (2012). Deconstructing the third dimension – How 3D culture microenvironments alter cellular cues. J Cell Sci 125, 3015-3024. doi:10.1242/jcs.079509
Balali-Mood, M., Naseri, K., Tahergorabi, Z. et al. (2021). Toxic mechanisms of five heavy metals: Mercury, lead, chromium, cadmium, and arsenic. Front Pharmacol 12, 643972. doi:10.3389/fphar.2021.643972
Beauchamp, P., Moritz, W., Kelm, J. M. et al. (2015). Development and characterization of a scaffold-free 3D spheroid model of induced pluripotent stem cell-derived human cardiomyocytes. Tissue Eng Part C Methods 21, 852-861. doi:10.1089/ten.tec.2014.0376
Beauchamp, P., Jackson, C. B., Ozhathil, L. C. et al. (2020). 3D co-culture of hiPSC-derived cardiomyocytes with cardiac fibroblasts improves tissue-like features of cardiac spheroids. Front Mol Biosci 7, 14. doi:10.3389/fmolb.2020.00014
Bell, S. M., Chang, X., Wambaugh, J. F. et al. (2018). In vitro to in vivo extrapolation for high throughput prioritization and decision making. Toxicol In Vitro 47, 213-227. doi:10.1016/j.tiv.2017.11.016
Berg, E. L., Hsu, Y.-C. and Lee, J. A. (2014). Consideration of the cellular microenvironment: Physiologically relevant co-culture systems in drug discovery. Adv Drug Deliv Rev 69, 190-204. doi:10.1016/j.addr.2014.01.013
Blanchette, A. D., Grimm, F. A., Dalaijamts, C. et al. (2019). Thorough QT/QTc in a dish: An in vitro human model that accurately predicts clinical concentration-QTc relationships. Clin Pharmacol Ther 105, 1175-1186. doi:10.1002/cpt.1259
Blanchette, A. D., Burnett, S. D., Grimm, F. A. et al. (2020). A Bayesian method for population-wide cardiotoxicity hazard and risk characterization using an in vitro human model. Toxicol Sci 178, 391-403. doi:10.1093/toxsci/kfaa151
Blanchette, A. D., Burnett, S. D., Rusyn, I. et al. (2022). A tiered approach to population-based in vitro testing for cardiotoxicity: Balancing estimates of potency and variability. J Pharmacol Toxicol Methods 114, 107154. doi:10.1016/j.vascn.2022.107154
Blinova, K., Stohlman, J., Vicente, J. et al. (2017). Comprehensive translational assessment of human-induced pluripotent stem cell derived cardiomyocytes for evaluating drug-induced arrhythmias. Toxicol Sci 155, 234-247. doi:10.1093/toxsci/kfw200
Blinova, K., Dang, Q., Millard, D. et al. (2018). International multisite study of human-induced pluripotent stem cell-derived cardiomyocytes for drug proarrhythmic potential assessment. Cell Rep 24, 3582-3592. doi:10.1016/j.celrep.2018.08.079
Bowes, J., Brown, A. J., Hamon, J. et al. (2012). Reducing safety-related drug attrition: The use of in vitro pharmacological profiling. Nat Rev Drug Discov 11, 909-922. doi:10.1038/nrd3845
Burnett, S. D., Blanchette, A. D., Grimm, F. A. et al. (2019). Population-based toxicity screening in human induced pluripotent stem cell-derived cardiomyocytes. Toxicol Appl Pharmacol 381, 114711. doi:10.1016/j.taap.2019.114711
Burnett, S. D., Blanchette, A. D., Chiu, W. A. et al. (2021a). Cardiotoxicity hazard and risk characterization of toxcast chemicals using human induced pluripotent stem cell-derived cardiomyocytes from multiple donors. Chem Res Toxicol 34, 2110-2124. doi:10.1021/acs.chemrestox.1c00203
Burnett, S. D., Blanchette, A. D., Chiu, W. A. et al. (2021b). Human induced pluripotent stem cell (iPSC)-derived cardiomyocytes as an in vitro model in toxicology: Strengths and weaknesses for hazard identification and risk characterization. Expert Opin Drug Metab Toxicol 17, 887-902. doi:10.1080/17425255.2021.1894122
Burridge, P. W., Li, Y. F., Matsa, E. et al. (2016). Human induced pluripotent stem cell-derived cardiomyocytes recapitulate the predilection of breast cancer patients to doxorubicin-induced cardiotoxicity. Nat Med 22, 547-556. doi:10.1038/nm.4087
Cai, S., Rao, X., Ye, J. et al. (2020). Relationship between urinary bisphenol a levels and cardiovascular diseases in the U.S. adult population, 2003-2014. Ecotoxicol Environ Saf 192, 110300. doi:10.1016/j.ecoenv.2020.110300
Chaudhari, U., Nemade, H., Gaspar, J. A. et al. (2016). MicroRNAs as early toxicity signatures of doxorubicin in human-induced pluripotent stem cell-derived cardiomyocytes. Arch Toxicol 90, 3087-3098. doi:10.1007/s00204-016-1668-0
Chaudhari, U., Nemade, H., Sureshkumar, P. et al. (2018). Functional cardiotoxicity assessment of cosmetic compounds using human-induced pluripotent stem cell-derived cardiomyocytes. Arch Toxicol 92, 371-381. doi:10.1007/s00204-017-2065-z
Chen, I. Y., Matsa, E. and Wu, J. C. (2016). Induced pluripotent stem cells: At the heart of cardiovascular precision medicine. Nat Rev Cardiol 13, 333-349. doi:10.1038/nrcardio.2016.36
Chen, Z., Lloyd, D., Zhou, Y.-H. et al. (2021). Risk characterization of environmental samples using in vitro bioactivity and polycyclic aromatic hydrocarbon concentrations data. Toxicol Sci 179, 108-120. doi:10.1093/toxsci/kfaa166
Chiu, W. A. and Rusyn, I. (2018). Advancing chemical risk assessment decision-making with population variability data: Challenges and opportunities. Mamm Genome 29, 182-189. doi:10.1007/s00335-017-9731-6
Clements, M. and Thomas, N. (2014). High-throughput multi-parameter profiling of electrophysiological drug effects in human embryonic stem cell derived cardiomyocytes using multi-electrode arrays. Toxicol Sci 140, 445-461. doi:10.1093/toxsci/kfu084
Clements, M., Millar, V., Williams, A. S. et al. (2015). Bridging functional and structural cardiotoxicity assays using human embryonic stem cell-derived cardiomyocytes for a more comprehensive risk assessment. Toxicol Sci 148, 241-260. doi:10.1093/toxsci/kfv180
Cosselman, K. E., Navas-Acien, A. and Kaufman, J. D. (2015). Environmental factors in cardiovascular disease. Nat Rev Cardiol 12, 627-642. doi:10.1038/nrcardio.2015.152
Daneshian, M., Kamp, H., Hengstler, J. et al. (2016). Highlight report: Launch of a large integrated European in vitro toxicology project: EU-ToxRisk. Arch Toxicol 90, 1021-1024. doi:10.1007/s00204-016-1698-7
da Rocha, A. M., Creech, J., Thonn, E. et al. (2020). Detection of drug-induced Torsades de Pointes arrhythmia mechanisms using hiPSC-CM syncytial monolayers in a high-throughput screening voltage sensitive dye assay. Toxicol Sci 173, 402-415. doi:10.1093/toxsci/kfz235
Dix, D. J., Houck, K. A., Martin, M. T. et al. (2007). The ToxCast program for prioritizing toxicity testing of environmental chemicals. Toxicol Sci 95, 5-12. doi:10.1093/toxsci/kfl103
Ferdinandy, P., Baczkó, I., Bencsik, P. et al. (2019). Definition of hidden drug cardiotoxicity: Paradigm change in cardiac safety testing and its clinical implications. Eur Heart J 40, 1771-1777. doi:10.1093/eurheartj/ehy365
Feric, N. T., Pallotta, I., Singh, R. et al. (2019). Engineered cardiac tissues generated in the Biowire™ II: A platform for human-based drug discovery. Toxicol Sci 172, 89-97. doi:10.1093/toxsci/kfz168
Feyen, D. A., McKeithan, W. L., Bruyneel, A. A. et al. (2020). Metabolic maturation media improve physiological function of human iPSC-derived cardiomyocytes. Cell Rep 32, 107925. doi:10.1016/j.celrep.2020.107925
Fischer, I., Milton, C. and Wallace, H. (2020). Toxicity testing is evolving! Toxicol Res 9, 67-80. doi:10.1093/toxres/tfaa011
Forsythe, S. D., Devarasetty, M., Shupe, T. et al. (2018). Environmental toxin screening using human-derived 3D bioengineered liver and cardiac organoids. Front Public Health 6, 103. doi:10.3389/fpubh.2018.00103
Frommeyer, G. and Eckardt, L. (2016). Drug-induced proarrhythmia: Risk factors and electrophysiological mechanisms. Nat Rev Cardiol 13, 36-47. doi:10.1038/nrcardio.2015.110
Gao, X. and Wang, H.-S. (2014). Impact of bisphenol a on the cardiovascular system-epidemiological and experimental evidence and molecular mechanisms. Int J Environ Res Public Health 11, 8399-8413. doi:10.3390/ijerph110808399
Garbern, J. C. and Lee, R. T. (2021). Mitochondria and metabolic transitions in cardiomyocytes: Lessons from development for stem cell-derived cardiomyocytes. Stem Cell Res Ther 12, 177. doi:10.1186/s13287-021-02252-6
Georgiadis, N., Tsarouhas, K., Tsitsimpikou, C. et al. (2018). Pesticides and cardiotoxicity. Where do we stand? Toxicol Appl Pharmacol 353, 1-14. doi:10.1016/j.taap.2018.06.004
Gintant, G. (2011). An evaluation of hERG current assay performance: Translating preclinical safety studies to clinical QT prolongation. Pharmacol Ther 129, 109-119. doi:10.1016/j.pharmthera.2010.08.008
Gintant, G., Sager, P. T. and Stockbridge, N. (2016). Evolution of strategies to improve preclinical cardiac safety testing. Nat Rev Drug Discov 15, 457-471. doi:10.1038/nrd.2015.34
Gintant, G., Burridge, P., Gepstein, L. et al. (2019). Use of human induced pluripotent stem cell-derived cardiomyocytes in preclinical cancer drug cardiotoxicity testing: A scientific statement from the american heart association. Circ Res 125, e75-e92. doi:10.1161/RES.0000000000000291
Gomez-Garcia, M. J., Quesnel, E., Al-Attar, R. et al. (2021). Maturation of human pluripotent stem cell derived cardiomyocytes in vitro and in vivo. Semin Cell Dev Biol 118, 163-171. doi:10.1016/j.semcdb.2021.05.022
Grimm, F. A., Iwata, Y., Sirenko, O. et al. (2015). High-content assay multiplexing for toxicity screening in induced pluripotent stem cell-derived cardiomyocytes and hepatocytes. Assay Drug Dev Technol 13, 529-546. doi:10.1089/adt.2015.659
Grimm, F. A., Blanchette, A., House, J. S. et al. (2018). A human population-based organotypic in vitro model for cardiotoxicity screening. ALTEX 35, 441-452. doi:10.14573/altex.1805301
Grimm, F. A., Klaren, W. D., Li, X. et al. (2020). Cardiovascular effects of polychlorinated biphenyls and their major metabolites. Environ Health Perspect 128, 077008. doi:10.1289/EHP7030
Guo, L., Coyle, L., Abrams, R. M. et al. (2013). Refining the human ipsc-cardiomyocyte arrhythmic risk assessment model. Toxicol Sci 136, 581-594. doi:10.1093/toxsci/kft205
Guth, B. D. (2007). Preclinical cardiovascular risk assessment in modern drug development. Toxicol Sci 97, 4-20. doi:10.1093/toxsci/kfm026
Hann, M. M. and Oprea, T. I. (2004). Pursuing the leadlikeness concept in pharmaceutical research. Curr Opin Chem Biol 8, 255-263. doi:10.1016/j.cbpa.2004.04.003
Hartung, T., Luechtefeld, T., Maertens, A. et al. (2013). Food for thought … integrated testing strategies for safety assessments. ALTEX 30, 3-18. doi:10.14573/altex.2013.1.003
Horvath, P., Aulner, N., Bickle, M. et al. (2016). Screening out irrelevant cell-based models of disease. Nat Rev Drug Discov 15, 751-769. doi:10.1038/nrd.2016.175
Hsieh, N.-H., Chen, Z., Rusyn, I. et al. (2021). Risk characterization and probabilistic concentration-response modeling of complex environmental mixtures using new approach methodologies (NAMs) data from organotypic in vitro human stem cell assays. Environ Health Perspect 129, 17004. doi:10.1289/EHP7600
Huebsch, N., Loskill, P., Deveshwar, N. et al. (2016). Miniaturized iPS-cell-derived cardiac muscles for physiologically relevant drug response analyses. Sci Rep 6, 24726. doi:10.1038/srep24726
ICCVAM (2018). A Strategic Roadmap for Establishing New Approaches to Evaluate the Safety of Chemicals and Medical Products in the United States. National Toxicology Program Research Triangle Park, NC. doi:10.22427/NTP-ICCVAM-ROADMAP2018
ICH (2001). S7A Safety Pharmacology Studies for Human Pharmaceuticals 36791-36792. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/s7a-safety-pharmacology-studies-human-pharmaceuticals
ICH (2005). S7B Non-Clinical Evaluation of the Potential for Delayed Ventricular Repolarization (QT Interval Prolongation) by Human Pharmaceuticals. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/s7b-nonclinical-evaluation-potential-delayed-ventricular-repolarization-qt-interval-prolongation
Incardona, J. P. (2017). Molecular mechanisms of crude oil developmental toxicity in fish. Arch Environ Contam Toxicol 73, 19-32. doi:10.1007/s00244-017-0381-1
Ingber, D. E. (2020). Is it time for reviewer 3 to request human organ chip experiments instead of animal validation studies? Adv Sci 7, 2002030. doi:10.1002/advs.202002030
Judson, R., Richard, A., Dix, D. J. et al. (2009). The toxicity data landscape for environmental chemicals. Environ Health Perspect 117, 685-695. doi:10.1289/ehp.0800168
Kleensang, A., Maertens, A., Rosenberg, M. et al. (2014). Pathways of toxicity. ALTEX 31, 53-61. doi:10.14573/altex.1309261
Kofron, C., Kim, T. Y., Munarin, F. et al. (2021). A predictive in vitro risk assessment platform for pro-arrhythmic toxicity using human 3D cardiac microtissues. Sci Rep 11, 10228. doi:10.1038/s41598-021-89478-9
Krewski, D., Acosta Jr, D., Andersen, M. et al. (2010). Toxicity testing in the 21st century: A vision and a strategy. J Toxicol Environ Health B Crit Rev 13, 51-138. doi:10.1080/10937404.2010.483176
Krishna, S., Berridge, B. and Kleinstreuer, N. (2020). High-throughput screening to identify chemical cardiotoxic potential. Chem Res Toxicol 34, 566-583. doi:10.1021/acs.chemrestox.0c00382
Krishna, S., Borrel, A., Huang, R. et al. (2022). High-throughput chemical screening and structure-based models to predict hERG inhibition. Biology 11, 209. doi:10.3390/biology11020209
Lee-Montiel, F. T., Laemmle, A., Dumont, L. et al. (2021). Integrated hiPSC-based liver and heart microphysiological systems predict unsafe drug-drug interaction. Front Pharmacol 12, 667010. doi:10.3389/fphar.2021.667010
Lee, E. K., Kurokawa, Y. K., Tu, R. et al. (2015). Machine learning plus optical flow: A simple and sensitive method to detect cardioactive drugs. Sci Rep 5, 11817. doi:10.1038/srep11817
Lind, L., Araujo, J. A., Barchowsky, A. et al. (2021). Key characteristics of cardiovascular toxicants. Environ Health Perspect 129, 95001. doi:10.1289/EHP9321
Lind, Y. S., Lind, P. M., Salihovic, S. et al. (2013). Circulating levels of persistent organic pollutants (POPs) are associated with left ventricular systolic and diastolic dysfunction in the elderly. Environ Res 123, 39-45. doi:10.1016/j.envres.2013.02.007
Lipinski, C. A., Lombardo, F., Dominy, B. W. et al. (1997). Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv Drug Deliv Rev 23, 3-25. doi:10.1016/S0169-409X(96)00423-1
Luo, Y.-S., Chen, Z., Blanchette, A. D. et al. (2021). Relationships between constituents of energy drinks and beating parameters in human induced pluripotent stem cell (iPSC)-derived cardiomyocytes. Food Chem Toxicol 149, 111979. doi:10.1016/j.fct.2021.111979
Magdy, T., Schuldt, A. J., Wu, J. C. et al. (2018). Human induced pluripotent stem cell (hiPSC)-derived cells to assess drug cardiotoxicity: Opportunities and problems. Annu Rev Pharmacol Toxicol 58, 83-103. doi:10.1146/annurev-pharmtox-010617-053110
Marris, C., Kompella, S. N., Miller, M. et al. (2020). Polyaromatic hydrocarbons in pollution: A heart‐breaking matter. J Physiol 598, 227-247. doi:10.1113/JP278885
Marx, U., Akabane, T., Andersson, T. B. et al. (2020). Biology-inspired microphysiological systems to advance patient benefit and animal welfare in drug development. ALTEX 37, 365-394. doi:10.14573/altex.2001241
Mathur, A., Loskill, P., Shao, K. et al. (2015). Human iPSC-based cardiac microphysiological system for drug screening applications. Sci Rep 5, 8883. doi:10.1038/srep08883
Meek, M., Boobis, A., Cote, I. et al. (2014). New developments in the evolution and application of the WHO/IPCs framework on mode of action/species concordance analysis. J Appl Toxicol 34, 1-18. doi:10.1002/jat.2949
Mercola, M., Colas, A. and Willems, E. (2013). Induced pluripotent stem cells in cardiovascular drug discovery. Circ Res 112, 534-548. doi:10.1161/CIRCRESAHA.111.250266
Meyer, T., Tiburcy, M. and Zimmermann, W.-H. (2019). Cardiac macrotissues-on-a-plate models for phenotypic drug screens. Adv Drug Deliv Rev 140, 93-100. doi:10.1016/j.addr.2019.03.002
Mikryukov, A. A., Mazine, A., Wei, B. et al. (2021). BMP10 signaling promotes the development of endocardial cells from human pluripotent stem cell-derived cardiovascular progenitors. Cell Stem Cell 28, 96-111.e7. doi:10.1016/j.stem.2020.10.003
Mironov, V., Trusk, T., Kasyanov, V. et al. (2009). Biofabrication: A 21st century manufacturing paradigm. Biofabrication 1, 022001. doi:10.1088/1758-5082/1/2/022001
Moffat, J. G., Vincent, F., Lee, J. A. et al. (2017). Opportunities and challenges in phenotypic drug discovery: An industry perspective. Nat Rev Drug Discov 16, 531-543. doi:10.1038/nrd.2017.111
NRC – National Research Council (2007). Toxicity Testing in the 21st Century: A Vision and a Strategy. Washington DC, USA: The National Academies Press. doi:10.17226/11970
Nerbonne, J. M., Nichols, C. G., Schwarz, T. L. et al. (2001). Genetic manipulation of cardiac K+ channel function in mice: What have we learned, and where do we go from here? Circ Res 89, 944-956. doi:10.1161/hh2301.100349
Nunes, S. S., Miklas, J. W., Liu, J. et al. (2013). Biowire: A platform for maturation of human pluripotent stem cell-derived cardiomyocytes. Nat Methods 10, 781-787. doi:10.1038/nmeth.2524
O’Hara, T. and Rudy, Y. (2012). Quantitative comparison of cardiac ventricular myocyte electrophysiology and response to drugs in human and nonhuman species. Am J Physiol Heart Circ Physiol 302, H1023-H1030. doi:10.1152/ajpheart.00785.2011
Olson, H., Betton, G., Robinson, D. et al. (2000). Concordance of the toxicity of pharmaceuticals in humans and in animals. Regul Toxicol Pharmacol 32, 56-67. doi:10.1006/rtph.2000.1399
Ong, C. S., Fukunishi, T., Zhang, H. et al. (2017). Biomaterial-free three-dimensional bioprinting of cardiac tissue using human induced pluripotent stem cell derived cardiomyocytes. Sci Rep 7, 4566. doi:10.1038/s41598-017-05018-4
Parish, S. T., Aschner, M., Casey, W. et al. (2020). An evaluation framework for new approach methodologies (NAMs) for human health safety assessment. Regul Toxicol Pharmacol 112, 104592. doi:10.1016/j.yrtph.2020.104592
Pauluhn, J. (2005). Overview of inhalation exposure techniques: Strengths and weaknesses. Exp Toxicol Pathol 57, 111-128. doi:10.1016/j.etp.2005.05.014
Pfeiffer-Kaushik, E. R., Smith, G. L., Cai, B. et al. (2019). Electrophysiological characterization of drug response in hSC-derived cardiomyocytes using voltage-sensitive optical platforms. J Pharmacol Toxicol Methods 99, 106612. doi:10.1016/j.vascn.2019.106612
Pointon, A., Pilling, J., Dorval, T. et al. (2017). From the cover: High-throughput imaging of cardiac microtissues for the assessment of cardiac contraction during drug discovery. Toxicol Sci 155, 444-457. doi:10.1093/toxsci/kfw227
Polonchuk, L., Chabria, M., Badi, L. et al. (2017). Cardiac spheroids as promising in vitro models to study the human heart microenvironment. Sci Rep 7, 7005. doi:10.1038/s41598-017-06385-8
Priest, B., Bell, I. M. and Garcia, M. (2008). Role of hERG potassium channel assays in drug development. Channels 2, 87-93. doi:10.4161/chan.2.2.6004
Protze, S. I., Lee, J. H. and Keller, G. M. (2019). Human pluripotent stem cell-derived cardiovascular cells: From developmental biology to therapeutic applications. Cell Stem Cell 25, 311-327. doi:10.1016/j.stem.2019.07.010
Prüss-Ustün, A. and Corvalán, C. F. (2006). Preventing Disease Through Healthy Environments: Towards an Estimate of the Environmental Burden of Disease. World Health Organization. https://apps.who.int/iris/handle/10665/43457
Rampoldi, A., Singh, M., Wu, Q. et al. (2019). Cardiac toxicity from ethanol exposure in human-induced pluripotent stem cell-derived cardiomyocytes. Toxicol Sci 169, 280-292. doi:10.1093/toxsci/kfz038
Ravenscroft, S. M., Pointon, A., Williams, A. W. et al. (2016). Cardiac non-myocyte cells show enhanced pharmacological function suggestive of contractile maturity in stem cell derived cardiomyocyte microtissues. Toxicol Sci 152, 99-112. doi:10.1093/toxsci/kfw069
Richards, D. J., Li, Y., Kerr, C. M. et al. (2020). Human cardiac organoids for the modelling of myocardial infarction and drug cardiotoxicity. Nat Biomed Eng 4, 446-462. doi:10.1038/s41551-020-0539-4
Sallam, K., Li, Y., Sager, P. T. et al. (2015). Finding the rhythm of sudden cardiac death: New opportunities using induced pluripotent stem cell-derived cardiomyocytes. Circ Res 116, 1989-2004. doi:10.1161/CIRCRESAHA.116.304494
Sarazan, R. D., Mittelstadt, S., Guth, B. et al. (2011). Cardiovascular function in nonclinical drug safety assessment: Current issues and opportunities. Int J Toxicol 30, 272-286. doi:10.1177/1091581811398963
Sauve-Ciencewicki, A., Davis, K. P., McDonald, J. et al. (2019). A simple problem formulation framework to create the right solution to the right problem. Regul Toxicol Pharmacol 101, 187-193. doi:10.1016/j.yrtph.2018.11.015
Schaaf, S., Shibamiya, A., Mewe, M. et al. (2011). Human engineered heart tissue as a versatile tool in basic research and preclinical toxicology. PLoS One 6, e26397. doi:10.1371/journal.pone.0026397
Schmid, C., Abi-Gerges, N., Leitner, M. G. et al. (2021). Ion channel expression and electrophysiology of singular human (primary and induced pluripotent stem cell-derived) cardiomyocytes. Cells 10, 3370. doi:10.3390/cells10123370
Scuderi, G. J. and Butcher, J. (2017). Naturally engineered maturation of cardiomyocytes. Front Cell Dev Biol 5, 50. doi:10.3389/fcell.2017.00050
Sevim, Ç., Doğan, E. and Comakli, S. (2020). Cardiovascular disease and toxic metals. Curr Opin Toxicol 19, 88-92. doi:10.1016/j.cotox.2020.01.004
Sharma, A., Burridge, P. W., McKeithan, W. L. et al. (2017). High-throughput screening of tyrosine kinase inhibitor cardiotoxicity with human induced pluripotent stem cells. Sci Transl Med 9, eaaf2584. doi:10.1126/scitranslmed.aaf2584
Sirenko, O., Cromwell, E. F., Crittenden, C. et al. (2013). Assessment of beating parameters in human induced pluripotent stem cells enables quantitative in vitro screening for cardiotoxicity. Toxicol Appl Pharmacol 273, 500-507. doi:10.1016/j.taap.2013.09.017
Sirenko, O., Grimm, F. A., Ryan, K. R. et al. (2017). In vitro cardiotoxicity assessment of environmental chemicals using an organotypic human induced pluripotent stem cell-derived model. Toxicol Appl Pharmacol 322, 60-74. doi:10.1016/j.taap.2017.02.020
Skardal, A., Murphy, S. V., Devarasetty, M. et al. (2017). Multi-tissue interactions in an integrated three-tissue organ-on-a-chip platform. Sci Rep 7, 8837. doi:10.1038/s41598-017-08879-x
Skardal, A., Aleman, J., Forsythe, S. et al. (2020). Drug compound screening in single and integrated multi-organoid body-on-a-chip systems. Biofabrication 12, 025017. doi:10.1088/1758-5090/ab6d36
Swinney, D. C. and Anthony, J. (2011). How were new medicines discovered? Nat Rev Drug Discov 10, 507-519. doi:10.1038/nrd3480
Swinney, D. C. (2013). Phenotypic vs. target‐based drug discovery for first‐in‐class medicines. Clin Pharmacol Ther 93, 299-301. doi:10.1038/clpt.2012.236
Takasuna, K., Asakura, K., Araki, S. et al. (2017). Comprehensive in vitro cardiac safety assessment using human stem cell technology: Overview of CSAHi HEART initiative. J Pharmacol Toxicol Methods 83, 42-54. doi:10.1016/j.vascn.2016.09.004
Teuschler, L., Klaunig, J., Carney, E. et al. (2002). Support of science-based decisions concerning the evaluation of the toxicology of mixtures: A new beginning. Regul Toxicol Pharmacol 36, 34-39. doi:10.1006/rtph.2002.1570
Törnqvist, E., Annas, A., Granath, B. et al. (2014). Strategic focus on 3R principles reveals major reductions in the use of animals in pharmaceutical toxicity testing. PLoS One 9, e101638. doi:10.1371/journal.pone.0101638
Truskey, G. A. (2018). Human microphysiological systems and organoids as in vitro models for toxicological studies. Front Public Health 6, 185. doi:10.3389/fpubh.2018.00185
Ulmer, B. M., Stoehr, A., Schulze, M. L. et al. (2018). Contractile work contributes to maturation of energy metabolism in hiPSC-derived cardiomyocytes. Stem Cell Rep 10, 834-847. doi:10.1016/j.stemcr.2018.01.039
Verheijen, M., Schrooders, Y., Gmuender, H. et al. (2018). Bringing in vitro analysis closer to in vivo: Studying doxorubicin toxicity and associated mechanisms in 3D human microtissues with PBPK-based dose modelling. Toxicol Lett 294, 184-192. doi:10.1016/j.toxlet.2018.05.029
Vincent, F., Loria, P., Pregel, M. et al. (2015). Developing predictive assays: The phenotypic screening “rule of 3”. Sci Transl Med 7, 293ps215-293ps215. doi:10.1126/scitranslmed.aab1201
Virani, S. S., Alonso, A., Benjamin, E. J. et al. (2020). Heart disease and stroke statistics – 2020 update: A report from the american heart association. Circulation 141, E139-E596. doi:10.1161/CIR.0000000000000757
Wambaugh, J. F., Wetmore, B. A., Pearce, R. et al. (2015). Toxicokinetic triage for environmental chemicals. Toxicol Sci 147, 55-67. doi:10.1093/toxsci/kfv118
Wetmore, B. A., Wambaugh, J. F., Ferguson, S. S. et al. (2013). Relative impact of incorporating pharmacokinetics on predicting in vivo hazard and mode of action from high-throughput in vitro toxicity assays. Toxicol Sci 132, 327-346. doi:10.1093/toxsci/kft012
Wetmore, B. A. (2015). Quantitative in vitro-to-in vivo extrapolation in a high-throughput environment. Toxicology 332, 94-101. doi:10.1016/j.tox.2014.05.012
Witty, A. D., Mihic, A., Tam, R. Y. et al. (2014). Generation of the epicardial lineage from human pluripotent stem cells. Nat Biotechnol 32, 1026-1035. doi:10.1038/nbt.3002
Wu, L., Rajamani, S., Li, H. et al. (2009). Reduction of repolarization reserve unmasks the proarrhythmic role of endogenous late Na+ current in the heart. Am J Physiol Heart Circ Physiol 297, H1048-H1057. doi:10.1152/ajpheart.00467.2009
Yang, F. and Massey, I. Y. (2019). Exposure routes and health effects of heavy metals on children. Biometals 32, 563-573. doi:10.1007/s10534-019-00193-5
Zhang, S., Zhou, Z., Gong, Q. et al. (1999). Mechanism of block and identification of the verapamil binding domain to herg potassium channels. Circ Res 84, 989-998. doi:10.1161/01.res.84.9.989
Zhao, Y., Rafatian, N., Wang, E. Y. et al. (2020). Towards chamber specific heart-on-a-chip for drug testing applications. Adv Drug Deliv Rev 165-166, 60-76. doi:10.1016/j.addr.2019.12.002
Zink, D., Chuah, J. K. C. and Ying, J. Y. (2020). Assessing toxicity with human cell-based in vitro methods. Trends Mol Med 26, 570-582. doi:10.1016/j.molmed.2020.01.008
Zuppinger, C. (2019). 3D cardiac cell culture: A critical review of current technologies and applications. Front Cardiovasc Med 6, 87. doi:10.3389/fcvm.2019.00087