Next-generation risk assessment of chemicals – Rolling out a human-centric testing strategy to drive 3R implementation: The RISK-HUNT3R project perspective
Main Article Content
Abstract
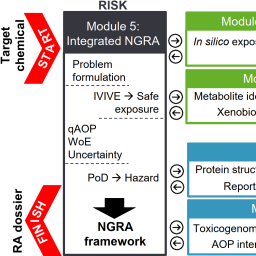
In many industrial sectors, there is a need for reliable ways to evaluate the safety of chemicals with methods anchored to human biology and pathology. For this purpose, many animal-free new approach methods (NAMs) have been developed and implemented in various stages of risk assessment. Now it is time to assemble individual NAMs into a comprehensive next-generation risk assessment (NGRA) strategy. The European Horizon 2020 RISK-HUNT3R project (Risk assessment of chemicals integrating human-centric next-generation testing strategies promoting the 3Rs) has been designed to promote a combination of computational toxicology, in vitro toxicology, and systems biology. It is anticipated that this approach will lead to faster and more accurate risk assessment procedures. The RISK-HUNT3R NGRA strategy will be developed to address the implementation of a comprehensive NAM toolbox into the regulatory framework. Critical conceptual approaches of the project include i) the integration of human-relevant data on biotransformation and elimination, ii) the translation of high-content mode-of-action datasets into predictions of adverse outcomes, iii) development of quantitative adverse outcome pathways (qAOPs), and iv) quantification of uncertainties associated with the predictions based on NGRA strategies. Many of the project steps will be used iteratively to generate datasets with sufficient quality and certainty for NGRA. Scientists and regulators will work together on case studies to evaluate practical applicability of NAMs and strategies to combine information therefrom. Here we delineate how the strategy will be deployed to establish an overall NGRA framework for chemicals, pesticides, food additives, and drugs.
Article Details

This work is licensed under a Creative Commons Attribution 4.0 International License.
Articles are distributed under the terms of the Creative Commons Attribution 4.0 International license (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution and reproduction in any medium, provided the original work is appropriately cited (CC-BY). Copyright on any article in ALTEX is retained by the author(s).
Bal-Price, A., Hogberg, H. T., Crofton, K. M. et al. (2018). Recommendation on test readiness criteria for new approach methods in toxicology: Exemplified for developmental neurotoxicity. ALTEX 35, 306-352. doi:10.14573/altex.1712081
Bloomingdale, P., Housand, C., Apgar, J. F. et al. (2017). Quantitative systems toxicology. Curr Opin Toxicol 4, 79-87. doi:10.1016/j.cotox.2017.07.003
Busquet, F., Kleensang, A., Rovida, C. et al. (2020). New European Union statistics on laboratory animal use – What really counts! ALTEX 37, 167-186. doi:10.14573/altex.2003241
Delp, J., Gutbier, S., Klima, S. et al. (2018). A high-throughput approach to identify specific neurotoxicants/ developmental toxicants in human neuronal cell function assays. ALTEX 35, 235-253. doi:10.14573/altex.1712182
Dent, M., Amaral, R. T., Da Silva, P. A. et al. (2018). Principles underpinning the use of new methodologies in the risk assessment of cosmetic ingredients. Comput Toxicol 7, 20-26. doi:10.1016/j.comtox.2018.06.001
EFSA (2018). Guidance on Uncertainty Analysis in Scientific Assessments. EFSA J 16, e05123. doi:10.2903/j.efsa.2018.5123
EFSA (2019). Guidance on Communication of Uncertainty in Scientific Assessments. EFSA J 17, e05520. doi:10.2903/j.efsa.2019.5520
Ennever, F. K. and Lave, L. B. (2003). Implications of the lack of accuracy of the lifetime rodent bioassay for predicting human carcinogenicity. Regul Toxicol Pharmacol 38, 52-57. doi:10.1016/s0273-2300(03)00068-0
Escher, S. E., Kamp, H., Bennekou, S. H. et al. (2019). Towards grouping concepts based on new approach methodologies in chemical hazard assessment: The read-across approach of the EU-ToxRisk project. Arch Toxicol 93, 3643-3667. doi:10.1007/s00204-019-02591-7
Escher, S. E., Aguayo-Orozco, A., Benfenati, E. et al. (2022). Integrate mechanistic evidence from new approach methodologies (NAMs) into a read-across assessment to characterise trends in shared mode of action. Toxicol In Vitro 79, 105269. doi:10.1016/j.tiv.2021.105269
Hartung, T. and Leist, M. (2008). Food for thought ... on the evolution of toxicology and the phasing out of animal testing. ALTEX 25, 91-102. doi:10.14573/altex.2008.2.91
Hartung, T. (2009). Toxicology for the twenty-first century. Nature 460, 208-212. doi:10.1038/460208a
Hartung, T. (2017). Thresholds of toxicological concern – Setting a threshold for testing below which there is little concern. ALTEX 34, 331-351. doi:10.14573/altex.1707011
Heusinkveld, H., Braakhuis, H., Gommans, R. et al. (2020). Towards a mechanism-based approach for the prediction of nongenotoxic carcinogenic potential of agrochemicals. Crit Rev Toxicol 50, 725-739. doi:10.1080/10408444.2020.1841732
Jacobs, M. N., Colacci, A., Corvi, R. et al. (2020). Chemical carcinogen safety testing: OECD expert group international consensus on the development of an integrated approach for the testing and assessment of chemical non-genotoxic carcinogens. Arch Toxicol 94, 2899-2923. doi:10.1007/s00204-020-02784-5
Krebs, A., van Vugt-Lussenburg, B. M. A., Waldmann, T. et al. (2020). The EU-ToxRisk method documentation, data processing and chemical testing pipeline for the regulatory use of new approach methods. Arch Toxicol 94, 2435-2461. doi:10.1007/s00204-020-02802-6
Krewski, D., Andersen, M. E., Tyshenko, M. G. et al. (2020). Toxicity testing in the 21st century: Progress in the past decade and future perspectives. Arch Toxicol 94, 1-58. doi:10.1007/s00204-019-02613-4
Leist, M., Hartung, T. and Nicotera, P. (2008). The dawning of a new age of toxicology. ALTEX 25, 103-114. doi:10.14573/altex.2008.2.103
Leist, M. and Hartung, T. (2013). Inflammatory findings on species extrapolations: Humans are definitely no 70-kg mice. Arch Toxicol 87, 563-567. doi:10.1007/s00204-013-1038-0
Luijten, M., Rorije, E., Sprong, R. C. et al. (2020). Practical application of next generation risk assessment of chemicals for human health. Chem Res Toxicol 33, 693-694. doi:10.1021/acs.chemrestox.0c00074
Moné, M. J., Pallocca, G., Escher, S. E. et al. (2020). Setting the stage for next-generation risk assessment with non-animal approaches: The EU-ToxRisk project experience. Arch Toxicol 94, 3581-3592. doi:10.1007/s00204-020-02866-4
NRC – National Research Council (2007). Toxicity Testing in the 21st Century: A Vision and a Strategy. Washington, DC, USA: The National Academies Press. doi:10.17226/11970
Nyffeler, J., Willis, C., Lougee, R. et al. (2020). Bioactivity screening of environmental chemicals using imaging-based high-throughput phenotypic profiling. Toxicol Appl Pharmacol 389, 114876. doi:10.1016/j.taap.2019.114876
Sachana, M., Willett, C., Pistollato, F. et al. (2021). The potential of mechanistic information organised within the AOP framework to increase regulatory uptake of the developmental neurotoxicity (DNT) in vitro battery of assays. Reprod Toxicol 103, 159-170. doi:10.1016/j.reprotox.2021.06.006
Solomon, K. R., Wilks, M. F., Bachman, A. et al. (2016). Problem formulation for risk assessment of combined exposures to chemicals and other stressors in humans. Crit Rev Toxicol 46, 835-844. doi:10.1080/10408444.2016.1211617
US EPA (2011). Exposure Factors Handbook (Final Report). U.S. Environmental Protection Agency, Washington, DC. EPA/600/R-09/052F
US EPA (2014). Framework for human health risk assessment to inform decision making. U.S. Environmental Protection Agency, Washington, DC. EPA/100/R-14/001
Van der Bles, A. M., van der Linden, S., Freeman, A. L. J. et al. (2019). Communicating uncertainty about facts, numbers and science. R Soc Open Sci 6, 181870. doi:10.1098/rsos.181870
Van der Stel, W., Carta, G., Eakins, J. et al. (2021). New approach methods (NAMs) supporting read-across: Two neurotoxicity AOP-based IATA case studies. ALTEX 38, 615-635. doi:10.14573/altex.2103051
Weaver, R. J., Blomme, E. A., Chadwick, A. E. et al. (2020). Managing the challenge of drug-induced liver injury: A roadmap for the development and deployment of preclinical predictive models. Nat Rev Drug Discov 19, 131-148. doi:10.1038/s41573-019-0048-x
Wink, S., Hiemstra, S., Huppelschoten, S. et al. (2014). Quantitative high content imaging of cellular adaptive stress response pathways in toxicity for chemical safety assessment. Chem Res Toxicol 27, 338-355. doi:10.1021/tx4004038