Applying a next generation risk assessment framework for skin sensitisation to inconsistent new approach methodology information
Main Article Content
Abstract
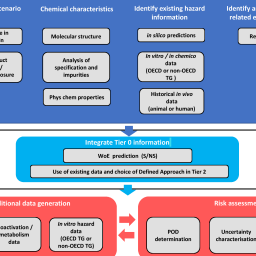
Cosmetic products must be safe for their intended use. Regulatory bans on animal testing for new ingredients have resulted in a shift towards the use of new approach methodologies (NAMs) such as in silico predictions and in chemico / in vitro data. Defined approaches (DAs) have been developed to interpret combinations of NAMs to provide information on skin sensitization hazard and potency, three having been adopted within OECD Test Guideline 497. However, the challenge remains as to how DAs can be used to derive a quantitative point of departure for use in next generation risk assessment (NGRA). Here we provide an update to our previously published NGRA framework and present two hypothetical consumer risk assessment scenarios (rinse-off and leave-on) on one case study ingredient. Diethanolamine (DEA) was selected as the case study ingredient based on the existing NAM information demonstrating differences with respect to the outcomes from in silico predictions and in chemico / in vitro data. Seven DAs were applied, and these differences resulted in divergent DA outcomes and reduced confidence with respect to the hazard potential and potency predictions. Risk assessment conclusion for the rinse-off exposure led to an overall decision of safe for all applied DAs. Risk assessment conclusion for the higher leave-on exposure was safe when based on some DAs but unsafe based on others. The reasons for this were evaluated as well as the inherent uncertainty from the use of each NAM and DA in the risk assessment, enabling further refinement of our NGRA framework.
Article Details

This work is licensed under a Creative Commons Attribution 4.0 International License.
Articles are distributed under the terms of the Creative Commons Attribution 4.0 International license (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution and reproduction in any medium, provided the original work is appropriately cited (CC-BY). Copyright on any article in ALTEX is retained by the author(s).
Anderson, S. E., Wells, J., Fedorowicz, A. et al. (2007). Evaluation of the contact and respiratory sensitization potential of volatile organic compounds generated by simulated indoor air chemistry. Toxicol Sci 97, 355-363. doi:10.1093/toxsci/kfm043
Api, A. M., Basketter, D. A., Cadby, P. A. et al. (2008). Dermal sensitization quantitative risk assessment (QRA) for fragrance ingredients. Regul Toxicol Pharmacol 52, 3-23. doi:10.1016/j.yrtph.2007.10.008
Api, A. M., Basketter, D., Bridges, J. et al. (2020). Updating exposure assessment for skin sensitization quantitative risk assessment for fragrance materials. Regul Toxicol Pharmacol 118, 104805. doi:10.1016/j.yrtph.2020.104805
Assaf Vandecasteele, H., Gautier, F., Tourneix, F. et al. (2021). Next generation risk assessment for skin sensitisation: A case study with propyl paraben. Regul Toxicol Pharmacol 123, 104936. doi:10.1016/j.yrtph.2021.104936
Basketter, D. A., Alépée, N., Ashikaga, T. et al. (2014). Categorization of chemicals according to their relative human skin sensitizing potency. Dermatitis 25, 11-21. doi:10.1097/der.0000000000000003
Basketter, D. and Safford, B. (2016). Skin sensitization quantitative risk assessment: A review of underlying assumptions. Regul Toxicol Pharmacol 74, 105-116. doi:10.1016/j.yrtph.2015.11.013
Brain, K. R., Walters, K. A., Green, D. M. et al. (2005). Percutaneous penetration of diethanolamine through human skin in vitro: Application from cosmetic vehicles. Food Chem Toxicol 43, 681-690. doi:10.1016/j.fct.2004.12.021
Chilton, M. L., Macmillan, D. S., Steger-Hartmann, T. et al. (2018). Making reliable negative predictions of human skin sensitisation using an in silico fragmentation approach. Regul Toxicol Pharmacol 95, 227-235. doi:10.1016/j.yrtph.2018.03.015
Cottrez, F., Boitel, E., Ourlin, J. C. et al. (2016). SENS-IS, a 3D reconstituted epidermis based model for quantifying chemical sensitization potency: Reproducibility and predictivity results from an inter-laboratory study. Toxicol In Vitro 32, 248-260. doi:10.1016/j.tiv.2016.01.007
Daniel, A. B., Strickland, J., Allen, D. et al. (2018). International regulatory requirements for skin sensitization testing. Regul Toxicol Pharmacol 95, 52-65. doi:10.1016/j.yrtph.2018.03.003
Del Bufalo, A., Pauloin, T., Alépée, N. et al. (2018). Alternative integrated testing for skin sensitization: Assuring consumer safety. Appl In Vitro Toxicol 4, 30-43. doi:10.1089/aivt.2017.0023
Desprez, B., Dent, M., Keller, D. et al. (2018). A strategy for systemic toxicity assessment based on non-animal approaches: The cosmetics Europe long range science strategy programme. Toxicol In Vitro 50, 137-146. doi:10.1016/j.tiv.2018.02.017
Enoch, S. J., Madden, J. C. and Cronin, M. T. (2008). Identification of mechanisms of toxic action for skin sensitisation using a smarts pattern based approach. SAR QSAR Environ Res 19, 555-578. doi:10.1080/10629360802348985
Ezendam, J., Braakhuis, H. M. and Vandebriel, R. J. (2016). State of the art in non-animal approaches for skin sensitization testing: From individual test methods towards testing strategies. Arch Toxicol 90, 2861-2883. doi:10.1007/s00204-016-1842-4
Fiume, M. M., Heldreth, B., Bergfeld, W. F. et al. (2017). Safety assessment of diethanolamine and its salts as used in cosmetics. Int J Toxicol 36, 89s-110s. doi:10.1177/1091581817707179
Gautier, F., Tourneix, F., Assaf Vandecasteele, H. et al. (2020). Read-across can increase confidence in the next generation risk assessment for skin sensitisation: A case study with resorcinol. Regul Toxicol Pharmacol 117, 104755. doi:10.1016/j.yrtph.2020.104755
Gerberick, G. F., Vassallo, J. D., Bailey, R. E. et al. (2004). Development of a peptide reactivity assay for screening contact allergens. Toxicol Sci 81, 332-343. doi:10.1093/toxsci/kfh213
GHS, U. (2021). Globally Harmonised System of Classification and Labelling of Chemicals (GHS). 9th revision.
Gilmour, N., Kern, P. S., Alépée, N. et al. (2020). Development of a next generation risk assessment framework for the evaluation of skin sensitisation of cosmetic ingredients. Regul Toxicol Pharmacol 116, 104721. doi:10.1016/j.yrtph.2020.104721
Gilmour, N., Reynolds, J., Przybylak, K. et al. (2022). Next generation risk assessment for skin allergy: Decision making using new approach methodologies. Regul Toxicol Pharmacol 131, 105159. doi:10.1016/j.yrtph.2022.105159
Gomes, C., Nocairi, H., Thomas, M. et al. (2012). Stacking prediction for a binary outcome. Compstat 2012, 271-282.
Gomes, C., Nocairi, H., Thomas, M. et al. (2014). A simple and robust scoring technique for binary classification. J Artif Intell Res 3, 52-58.
Goodman, S. N. (1999). Toward evidence-based medical statistics. 2: The Bayes factor. Ann Intern Med 130, 1005-1013. doi:10.7326/0003-4819-130-12-199906150-00019
Griem, P., Goebel, C. and Scheffler, H. (2003). Proposal for a risk assessment methodology for skin sensitization based on sensitization potency data. Regul Toxicol Pharmacol 38, 269-290. doi:10.1016/j.yrtph.2003.07.001
Hirota, M., Ashikaga, T. and Kouzuki, H. (2018). Development of an artificial neural network model for risk assessment of skin sensitization using human cell line activation test, direct peptide reactivity assay, KeratinoSens™ and in silico structure alert parameter. J Appl Toxicol 38, 514-526. doi:10.1002/jat.3558
Hoffmann, S., Kleinstreuer, N., Alépée, N. et al. (2018). Non-animal methods to predict skin sensitization (I): The cosmetics Europe database. Crit Rev Toxicol 48, 344-358. doi:10.1080/10408444.2018.1429385
Hoffmann, S., Alépée, N., Gilmour, N. et al. (2022). Expansion of the cosmetics Europe skin sensitisation database with new substances and PPRA data. Regul Toxicol Pharmacol 131, 105169. doi:10.1016/j.yrtph.2022.105169
Jaworska, J. S., Natsch, A., Ryan, C. et al. (2015). Bayesian integrated testing strategy (ITS) for skin sensitization potency assessment: A decision support system for quantitative weight of evidence and adaptive testing strategy. Arch Toxicol 89, 2355-2383. doi:10.1007/s00204-015-1634-2
Kleinstreuer, N. C., Hoffmann, S., Alépée, N. et al. (2018). Non-animal methods to predict skin sensitization (II): An assessment of defined approaches. Crit Rev Toxicol 48, 359-374. doi:10.1080/10408444.2018.1429386
Kraeling, M. E., Yourick, J. J. and Bronaugh, R. L. (2004). In vitro human skin penetration of diethanolamine. Food Chem Toxicol 42, 1553-1561. doi:10.1016/j.fct.2004.04.016
Lessmann, H., Uter, W., Schnuch, A. et al. (2009). Skin sensitizing properties of the ethanolamines mono-, di-, and triethanolamine. Data analysis of a multicentre surveillance network (IVDK) and review of the literature. Contact Dermatitis 60, 243-255. doi:10.1111/j.1600-0536.2009.01506.x
Macmillan, D. S., Chilton, M. L., Gao, Y. et al. (2022). How to resolve inconclusive predictions from defined approaches for skin sensitisation in OECD Guideline No. 497. Regul Toxicol Pharmacol 135, 105248. doi:10.1016/j.yrtph.2022.105248
Natsch, A. and Emter, R. (2016). Nrf2 activation as a key event triggered by skin sensitisers: The development of the stable KeratinoSens reporter gene assay. Altern Lab Anim 44, 443-451. doi:10.1177/026119291604400513
Natsch, A., Emter, R., Haupt, T. et al. (2018). Deriving a no expected sensitization induction level for fragrance ingredients without animal testing: An integrated approach applied to specific case studies. Toxicol Sci 165, 170-185. doi:10.1093/toxsci/kfy135
Nocairi, H., Gomes, C., Thomas, M. et al. (2016). Improving stacking methodology for combining classifiers: Applications to cosmetic industry. Electronic Journal of Applied Statistical Analysis 9, 340-361. doi:10.1285/i20705948v9n2p340
OECD (2013). Test No. 122: Determination of pH, Acidity and Alkalinity. OECD Guidelines for the Testing of Chemicals, Section 1. OECD Publishing, Paris. doi:10.1787/9789264203686-en
OECD (2014). The Adverse Outcome Pathway for Skin Sensitisation Initiated by Covalent Binding to Proteins. OECD Series on Testing and Assessment, No. 168. OECD Publishing, Paris. doi:10.1787/9789264221444-en
OECD (2018). Test No. 442D: In Vitro Skin Sensitisation: ARE-Nrf2 Luciferase Test Method. OECD Guidelines for the Testing of Chemicals, Section 1. OECD Publishing, Paris. doi:10.1787/9789264229822-en
OECD (2020). Test No. 442C: In Chemico Skin Sensitisation: Assays addressing the Adverse Outcome Pathway key event on covalent binding to proteins. OECD Guidelines for the Testing of Chemicals, Section 4. OECD Publishing, Paris. doi:10.1787/9789264229709-en
OECD (2021). Guideline No. 497: Defined Approaches On Skin Sensitisation. OECD Guidelines for the Testing of Chemicals, Section 4. OECD Publishing, Paris. doi:10.1787/b92879a4-en
OECD (2022). Test No. 442E: In Vitro Skin Sensitisation. In Vitro Skin Sensitisation assays addressing the Key Event on activation of dendritic cells on the Adverse Outcome Pathway for Skin Sensitisation. OECD Guidelines for the Testing of Chemicals, Section 4. OECD Publishing, Paris. doi:10.1787/9789264264359-en
Patlewicz, G., Dimitrov, S. D., Low, L. K. et al. (2007). TIMES-SS – A promising tool for the assessment of skin sensitization hazard. A characterization with respect to the OECD validation principles for (Q)SARs and an external evaluation for predictivity. Regul Toxicol Pharmacol 48, 225-239. doi:10.1016/j.yrtph.2007.03.003
Patlewicz, G., Kuseva, C., Mehmed, A. et al. (2014). TIMES-SS – Recent refinements resulting from an industrial skin sensitisation consortium. SAR QSAR Environ Res 25, 367-391. doi:10.1080/1062936x.2014.900520
Patlewicz, G., Casati, S., Basketter, D. A. et al. (2016). Can currently available non-animal methods detect pre and pro-haptens relevant for skin sensitization? Regul Toxicol Pharmacol 82, 147-155. doi:10.1016/j.yrtph.2016.08.007
Piroird, C., Ovigne, J. M., Rousset, F. et al. (2015). The myeloid U937 skin sensitization test (U-SENS) addresses the activation of dendritic cell event in the adverse outcome pathway for skin sensitization. Toxicol In Vitro 29, 901-916. doi:10.1016/j.tiv.2015.03.009
Reisinger, K., Hoffmann, S., Alépée, N. et al. (2015). Systematic evaluation of non-animal test methods for skin sensitisation safety assessment. Toxicol In Vitro 29, 259-270. doi:10.1016/j.tiv.2014.10.018
Reynolds, J., Gilmour, N., Baltazar, M. T. et al. (2022). Decision making in next generation risk assessment for skin allergy: Using historical clinical experience to benchmark risk. Regul Toxicol Pharmacol 134, 105219. doi:10.1016/j.yrtph.2022.105219
Reynolds, G. R. J., Gilmour, N., Cubberley, R. et al. (submitted). A skin sensitisation next generation risk assessment case study for coumarin in hypothetical cosmetic products.
Robinson, M. K., Gerberick, G. F., Ryan, C. A. et al. (2000). The importance of exposure estimation in the assessment of skin sensitization risk. Contact Dermatitis 42, 251-259. doi:10.1034/j.1600-0536.2000.042005251.x
Sakaguchi, H., Ryan, C., Ovigne, J. M. et al. (2010). Predicting skin sensitization potential and inter-laboratory reproducibility of a human cell line activation test (h-CLAT) in the European cosmetics association (COLIPA) ring trials. Toxicol In Vitro 24, 1810-1820. doi:10.1016/j.tiv.2010.05.012
Spicer, C. W., Gordon, S. M., Holdren, M. W. et al. (2002). Hazardous Air Pollutant Handbook: Measurements, Properties, and Fate in Ambient Air. Boca Raton, FL, USA: CRC Press. doi:10.1201/9781420032352
Tollefsen, K. E., Scholz, S., Cronin, M. T. et al. (2014). Applying adverse outcome pathways (AOPs) to support integrated approaches to testing and assessment (IATA). Regul Toxicol Pharmacol 70, 629-640. doi:10.1016/j.yrtph.2014.09.009
Urbisch, D., Becker, M., Honarvar, N. et al. (2016). Assessment of pre- and pro-haptens using nonanimal test methods for skin sensitization. Chem Res Toxicol 29, 901-913. doi:10.1021/acs.chemrestox.6b00055
van Vliet, E., Kühnl, J., Goebel, C. et al. (2018). State-of-the-art and new options to assess T cell activation by skin sensitizers: Cosmetics Europe workshop. ALTEX 35, 179-192. doi:10.14573/altex.1709011
Wolpert, D. H. (1992). Stacked generalization. Neural Networks 5, 241-259.
Zang, Q., Mansouri, K., Williams, A. J. et al. (2017). In silico prediction of physicochemical properties of environmental chemicals using molecular fingerprints and machine learning. J Chem Inf Model 57, 36-49. doi:10.1021/acs.jcim.6b00625