A human iPSC-based in vitro neural network formation assay to investigate neurodevelopmental toxicity of pesticides
Main Article Content
Abstract
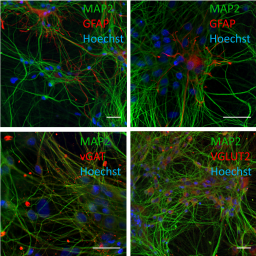
Proper brain development is based on the orchestration of key neurodevelopmental processes (KNDP), including the formation and function of neural networks. If at least one KNDP is affected by a chemical, an adverse outcome is expected. To enable a higher testing throughput than the guideline animal experiments, a developmental neurotoxicity (DNT) in vitro testing battery (DNT IVB) comprising a variety of assays that model several KNDPs was set up. Gap analysis revealed the need for a human-based assay to assess neural network formation and function (NNF). Therefore, we established the human NNF (hNNF) assay. A co-culture comprised of human induced pluripotent stem cell (hiPSC)-derived excitatory and inhibitory neurons as well as primary human astroglia was differentiated for 35 days on microelectrode arrays (MEA), and spontaneous electrical activity, together with cytotoxicity, was assessed on a weekly basis after washout of the compounds 24 h prior to measurements. In addition to the characterization of the test system, the assay was challenged with 28 compounds, mainly pesticides, identifying their DNT potential by evaluating specific spike-, burst-, and network parameters. This approach confirmed the suitability of the assay for screening environmental chemicals. Comparison of benchmark concentrations (BMC) with an NNF in vitro assay (rNNF) based on primary rat cortical cells revealed differences in sensitivity. Together with the successful implementation of hNNF data into a postulated stressor-specific adverse outcome pathway (AOP) network associated with a plausible molecular initiating event for deltamethrin, this study suggests the hNNF assay as a useful complement to the DNT IVB.
Article Details

This work is licensed under a Creative Commons Attribution 4.0 International License.
Articles are distributed under the terms of the Creative Commons Attribution 4.0 International license (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution and reproduction in any medium, provided the original work is appropriately cited (CC-BY). Copyright on any article in ALTEX is retained by the author(s).
Aschner, M., Ceccatelli, S., Daneshian, M. et al. (2017). Reference compounds for alternative test methods to indicate developmental neurotoxicity (DNT) potential of chemicals: Example lists and criteria for their selection and use. ALTEX 34, 49-74. doi:10.14573/altex.1604201
Bal-Price, A., Crofton, K. M., Leist, M. et al. (2015). International STakeholder NETwork (ISTNET): Creating a developmental neurotoxicity (DNT) testing road map for regulatory purposes. Arch Toxicol 89, 269-287. doi:10.1007/S00204-015-1464-2
Bal-Price, A., Hogberg, H. T., Crofton, K. M. et al. (2018). Recommendation on test readiness criteria for new approach methods in toxicology: Exemplified for developmental neurotoxicity. ALTEX 35, 306-352. doi:10.14573/altex.1712081
Bartmann, K., Hartmann, J., Kapr, J. et al. (2021). Measurement of electrical activity of differentiated human iPSC-derived neurospheres recorded by microelectrode arrays (MEA). In J. Llorens and M. Barenys (eds), Experimental Neurotoxicology Methods (473-488). Neuromethods 172. New York: Humana. doi:10.1007/978-1-0716-1637-6_22
Baumann, J., Barenys, M., Gassmann, K. et al. (2014). Comparative human and rat “neurosphere assay” for developmental neurotoxicity testing. Curr Protoc Toxicol 59, 12.21.1-12.21.24. doi:10.1002/0471140856.TX1221S59
Baumann, J., Dach, K., Barenys, M. et al. (2015). Application of the neurosphere assay for DNT hazard assessment: Challenges and limitations. In Methods in Pharmacology and Toxicology (29). Totowa, NJ: Humana Press. doi:10.1007/7653_2015_49
Baumann, J., Gassmann, K., Masjosthusmann, S. et al. (2016). Comparative human and rat neurospheres reveal species differences in chemical effects on neurodevelopmental key events. Arch Toxicol 90, 1415-1427. doi:10.1007/S00204-015-1568-8
Bayer, S. A., Altman, J., Russo, R. J. et al. (1993). Timetables of neurogenesis in the human brain based on experimentally determined patterns in the rat. Neurotoxicology 14, 83-144.
Bennett, D., Bellinger, D. C., Birnbaum, L. S. et al. (2016). Project TENDR: Targeting environmental neuro-developmental risks the TENDR consensus statement. Environ Health Perspect 124, A118-A122. doi:10.1289/EHP358
Bjørling-Poulsen, M., Andersen, H. R. and Grandjean, P. (2008). Potential developmental neurotoxicity of pesticides used in Europe. Environ Health 7, 50. doi:10.1186/1476-069X-7-50
Blum, J., Masjosthusmann, S., Bartmann, K. et al. (2022). Establishment of a human cell-based in vitro battery to assess developmental neurotoxicity hazard of chemicals. Chemosphere 311, 137035. doi:10.1016/J.chemosphere.2022.137035
Brown, J. P., Hall, D., Frank, C. L. et al. (2016). Evaluation of a microelectrode array-based assay for neural network ontogeny using training set chemicals. Toxicol Sci 154, 126-139. doi:10.1093/toxsci/kfw147
Carstens, K. E., Carpenter, A. F., Martin, M. M. et al. (2022). Integrating data from in vitro new approach methodologies for developmental neurotoxicity. Toxicol Sci 187, 62-79. doi:10.1093/toxsci/kfac018
Coecke, S., Goldberg, A. M., Allen, S. et al. (2007). Workgroup report: Incorporating in vitro alternative methods for developmental neurotoxicity into international hazard and risk assessment strategies. Environ Health Perspect 115, 924-931. doi:10.1289/ehp.9427
Crofton, K. M., Mundy, W. R., Lein, P. J. et al. (2011). Developmental neurotoxicity testing: Recommendations for developing alternative methods for the screening and prioritization of chemicals. ALTEX 28, 9-15. doi:10.14573/altex.2011.1.009
Crofton, K. M., Mundy, W. R. and Shafer, T. J. (2012). Developmental neurotoxicity testing: A path forward. Congenit Anom 52, 140-146. doi:10.1111/j.1741-4520.2012.00377.x
Crofton, K., Fritsche, E., Ylikomi, T. et al. (2014). International STakeholder NETwork (ISTNET) for creating a developmental neurotoxicity testing (DNT) roadmap for regulatory purposes. ALTEX 31, 223-224. doi:10.14573/altex.1402121
Crofton, K. M. and Mundy, W. R. (2021). External scientific report on the interpretation of data from the developmental neurotoxicity in vitro testing assays for use in integrated approaches for testing and assessment. EFSA Support Publ 18, 6924E. doi:10.2903/sp.efsa.2021.en-6924
Dach, K., Bendt, F., Huebenthal, U. et al. (2017). BDE-99 impairs differentiation of human and mouse NPCs into the oligodendroglial lineage by species-specific modes of action. Sci Rep 7, 44861. doi:10.1038/srep44861
Das, K. P., Freudenrich, T. M. and Mundy, W. R. (2004). Assessment of PC12 cell differentiation and neurite growth: A comparison of morphological and neurochemical measures. Neurotoxicol Teratol 26, 397-406. doi:10.1016/j.ntt.2004.02.006
Di Consiglio, E., Pistollato, F., Mendoza-De Gyves, E. et al. (2020). Integrating biokinetics and in vitro studies to evaluate developmental neurotoxicity induced by chlorpyrifos in human iPSC-derived neural stem cells undergoing differentiation towards neuronal and glial cells. Reprod Toxicol 98, 174-188. doi:10.1016/j.reprotox.2020.09.010
Doretto, S., Malerba, M., Ramos, M. et al. (2011). Oligodendrocytes as regulators of neuronal networks during early postnatal development. PLoS One 6, 19849. doi:10.1371/journal.pone.0019849
Druwe, I., Freudenrich, T. M., Wallace, K. et al. (2015). Sensitivity of neuroprogenitor cells to chemical-induced apoptosis using a multiplexed assay suitable for high-throughput screening. Toxicology 333, 14-24. doi:10.1016/J.TOX.2015.03.011
Druwe, I., Freudenrich, T. M., Wallace, K. et al. (2016). Comparison of human induced pluripotent stem cell-derived neurons and rat primary cortical neurons as in vitro models of neurite outgrowth. Appl Vitr Toxicol 2, 26-36. doi:10.1089/aivt.2015.0025
Dubois, J., Benders, M., Borradori-Tolsa, C. et al. (2008). Primary cortical folding in the human newborn: An early marker of later functional development. Brain 131, 2028-2041. doi:10.1093/BRAIN/AWN137
EFSA (2013). Scientific Opinion on the developmental neurotoxicity potential of acetamiprid and imidacloprid. EFSA J 11, 3471. doi:10.2903/j.efsa.2013.3471
Frank, C. L., Brown, J. P., Wallace, K. et al. (2017). Developmental neurotoxicants disrupt activity in cortical networks on microelectrode arrays: Results of screening 86 compounds during neural network formation. Toxicol Sci 160, 121-135. doi:10.1093/toxsci/kfx169
Fritsche, E., Alm, H., Baumann, J. et al. (2015). Literature review on in vitro and alternative developmental neurotoxicity (DNT) testing methods. EFSA Support Publ 12, 778E. doi:10.2903/sp.efsa.2015.en-778
Fritsche, E. (2017). Report on Integrated Testing Strategies for the identification and evaluation of chemical hazards associated with the developmental neurotoxicity (DNT), to facilitate discussions at the Joint EFSA/OECD Workshop on DNT. EFSA Support Publ 14, 1191E. doi:10.2903/sp.efsa.2017.en-1191
Fritsche, E., Crofton, K. M., Hernandez, A. F. et al. (2017). OECD/EFSA workshop on developmental neurotoxicity (DNT): The use of non-animal test methods for regulatory purposes. ALTEX 34, 311-315. doi:10.14573/altex.1701171
Fritsche, E., Barenys, M., Klose, J. et al. (2018a). Current availability of stem cell-based in vitro methods for developmental neurotoxicity (DNT) testing. Toxicol Sci 165, 21-30. doi:10.1093/toxsci/kfy178
Fritsche, E., Grandjean, P., Crofton, K. M. et al. (2018b). Consensus statement on the need for innovation, transition and implementation of developmental neurotoxicity (DNT) testing for regulatory purposes. Toxicol Appl Pharmacol 354, 3-6. doi:10.1016/J.TAAP.2018.02.004
Glantz, L. A., Gilmore, J. H., Hamer, R. M. et al. (2007). Synaptophysin and PSD-95 in the human prefrontal cortex from mid-gestation into early adulthood. Neuroscience 149, 582-591. doi:10.1016/j.neuroscience.2007.06.036
Goldman, L. R. and Koduru, S. (2000). Chemicals in the environment and developmental toxicity to children: A public health and policy perspective. Environ Health Perspect 108, 443-448. doi:10.1289/EHP.00108S3443
Grandjean, P. and Landrigan, P. (2006). Developmental neurotoxicity of industrial chemicals. Lancet 368, 2167-2178. doi:10.1016/S0140-6736(06)69665-7
Hama, H., Hara, C., Yamaguchi, K. et al. (2004). PKC signaling mediates global enhancement of excitatory synaptogenesis in neurons triggered by local contact with astrocytes. Neuron 41, 405-415. doi:10.1016/S0896-6273(04)00007-8
Hardy, A., Benford, D., Halldorsson, T. et al. (2017). Guidance on the use of the weight of evidence approach in scientific assessments. EFSA J 15, e04971. doi:10.2903/j.efsa.2017.4971
Harrill, J. A., Freudenrich, T. M., Machacek, D. W. et al. (2010). Quantitative assessment of neurite outgrowth in human embryonic stem cell-derived hN2 cells using automated high-content image analysis. Neurotoxicology 31, 277-290. doi:10.1016/j.neuro.2010.02.003
Harrill, J. A., Robinette, B. L. and Mundy, W. R. (2011). Use of high content image analysis to detect chemical-induced changes in synaptogenesis in vitro. Toxicol In Vitro 25, 368-387. doi:10.1016/j.tiv.2010.10.011
Harrill, J. A., Robinette, B. L., Freudenrich, T. et al. (2013). Use of high content image analyses to detect chemical-mediated effects on neurite sub-populations in primary rat cortical neurons. Neurotoxicology 34, 61-73. doi:10.1016/j.neuro.2012.10.013
Harrill, J. A., Freudenrich, T., Wallace, K. et al. (2018). Testing for developmental neurotoxicity using a battery of in vitro assays for key cellular events in neurodevelopment. Toxicol Appl Pharmacol 354, 24-39. doi:10.1016/J.TAAP.2018.04.001
Hernández‐Jerez, A., Adriaanse, P., Aldrich, A. et al. (2021). Development of Integrated Approaches to Testing and Assessment (IATA) case studies on developmental neurotoxicity (DNT) risk assessment. EFSA J 19, e06599. doi:10.2903/j.efsa.2021.6599
Hoelting, L., Klima, S., Karreman, C. et al. (2016). Stem cell-derived immature human dorsal root ganglia neurons to identify peripheral neurotoxicants. Stem Cells Transl Med 5, 476-487. doi:10.5966/SCTM.2015-0108
Hofrichter, M., Nimtz, L., Tigges, J. et al. (2017). Comparative performance analysis of human iPSC-derived and primary neural progenitor cells (NPC) grown as neurospheres in vitro. Stem Cell Res 25, 72-82. doi:10.1016/J.SCR.2017.10.013
Holst, C. B., Brøchner, C. B., Vitting-Seerup, K. et al. (2019). Astrogliogenesis in human fetal brain: Complex spatiotemporal immunoreactivity patterns of GFAP, S100, AQP4 and YKL-40. J Anat 235, 590-615. doi:10.1111/JOA.12948
Hyvärinen, T., Hyysalo, A., Kapucu, F. E. et al. (2019). Functional characterization of human pluripotent stem cell-derived cortical networks differentiated on laminin-521 substrate: Comparison to rat cortical cultures. Sci Rep 9, 17125. doi:10.1038/s41598-019-53647-8
Ishii, M. N., Yamamoto, K., Shoji, M. et al. (2017). Human induced pluripotent stem cell (hiPSC)-derived neurons respond to convulsant drugs when co-cultured with hiPSC-derived astrocytes. Toxicology 389, 130-138. doi:10.1016/j.tox.2017.06.010
Johnstone, A. F. M., Gross, G. W., Weiss, D. G. et al. (2010). Microelectrode arrays: A physiologically based neurotoxicity testing platform for the 21st century. Neurotoxicology 31, 331-350. doi:10.1016/j.neuro.2010.04.001
Johnston, G. A. R. (2013). Advantages of an antagonist: Bicuculline and other GABA antagonists. Br J Pharmacol 169, 328-336. doi:10.1111/BPH.12127
Karlin, A. (2002). Emerging structure of the nicotinic acetylcholine receptors. Nat Rev Neurosci 3, 102-114. doi:10.1038/nrn731
Klose, J., Tigges, J., Masjosthusmann, S. et al. (2021). TBBPA targets converging key events of human oligodendrocyte development resulting in two novel AOPs. ALTEX 38, 215-234. doi:10.14573/altex.2007201
Koch, K., Bartmann, K., Hartmann, J. et al. (2022). Scientific validation of human neurosphere assays for developmental neurotoxicity evaluation. Front Toxicol 4, 816370. doi:10.3389/ftox.2022.816370
Kosnik, M. B., Strickland, J. D., Marvel, S. W. et al. (2020). Concentration-response evaluation of ToxCast compounds for multivariate activity patterns of neural network function. Arch Toxicol 94, 469-484. doi:10.1007/s00204-019-02636-x
Kramer, N. I., Di Consiglio, E., Blaauboer, B. J. et al. (2015). Biokinetics in repeated-dosing in vitro drug toxicity studies. Toxicol In Vitro 30, 217-224. doi:10.1016/j.tiv.2015.09.005
Krug, A. K., Balmer, N. V., Matt, F. et al. (2013). Evaluation of a human neurite growth assay as specific screen for developmental neurotoxicants. Arch Toxicol 87, 2215-2231. doi:10.1007/s00204-013-1072-y
Kuehn, B. M. (2010). Increased risk of ADHD associated with early exposure to pesticides, PCBs. JAMA 304, 27-28. doi:10.1001/jama.2010.860
Leist, M. and Hartung, T. (2013). Reprint: Inflammatory findings on species extrapolations: Humans are definitely no 70-kg mice. ALTEX 30, 227-230. doi:10.14573/altex.2013.2.227
Leonzino, M., Busnelli, M., Antonucci, F. et al. (2016). The timing of the excitatory-to-inhibitory GABA switch is regulated by the oxytocin receptor via KCC2. Cell Rep 15, 96-103. doi:10.1016/j.celrep.2016.03.013
Little, D., Ketteler, R., Gissen, P. et al. (2019). Using stem cell-derived neurons in drug screening for neurological diseases. Neurobiol Aging 78, 130-141. doi:10.1016/j.neurobiolaging.2019.02.008
Maccioni, R. B. and Cambiazo, V. (1995). Role of microtubule-associated proteins in the control of microtubule assembly. Phys Rev 75, 835-864. doi:10.1152/physrev.1995.75.4.835
Macdonald, R. L., Rogers, C. J. and Twyman, R. E. (1989). Kinetic properties of the GABAA receptor main conductance state of mouse spinal cord neurones in culture. J Physiol 410, 479-499. doi:10.1113/jphysiol.1989.sp017545
Mack, C. M., Lin, B. J., Turner, J. D. et al. (2014). Burst and principal components analyses of MEA data for 16 chemicals describe at least three effects classes. Neurotoxicology 40, 75-85. doi:10.1016/j.neuro.2013.11.008
Martin, M. M., Baker, N. C., Boyes, W. K. et al. (2022). An expert-driven literature review of “negative” chemicals for developmental neurotoxicity (DNT) in vitro assay evaluation. Neurotoxicol Teratol 93, 107117. doi:10.1016/j.ntt.2022.107117
Masjosthusmann, S., Becker, D., Petzuch, B. et al. (2018). A transcriptome comparison of time-matched developing human, mouse and rat neural progenitor cells reveals human uniqueness. Toxicol Appl Pharmacol 354, 40-55. doi:10.1016/j.taap.2018.05.009
Masjosthusmann, S., Blum, J., Bartmann, K. et al. (2020). Establishment of an a priori protocol for the implementation and interpretation of an in‐vitro testing battery for the assessment of developmental neurotoxicity. EFSA Support Publ 17, 1938E. doi:10.2903/sp.efsa.2020.en-1938
Missale, C., Nash, S. R., Robinson, S. W. et al. (1998). Dopamine receptors: From structure to function. Physiol Rev 78, 189-225. doi:10.1152/physrev.1998.78.1.189
Miyamoto, A., Wake, H., Ishikawa, A. W. et al. (2016). Microglia contact induces synapse formation in developing somatosensory cortex. Nat Commun 7, 12540. doi:10.1038/ncomms12540
Neuman, R. S., Ben-Ari, Y., Gho, M. et al. (1988). Blockade of excitatory synaptic transmission by 6-cyano-7-nitroquinoxaline-2,3-dione (CNQX) in the hippocampus in vitro. Neurosci Lett 92, 64-68. doi:10.1016/0304-3940(88)90743-4
Nimtz, L., Klose, J., Masjosthusmann, S. et al. (2019). The neurosphere assay as an in vitro method for developmental neurotoxicity (DNT) evaluation. Neuromethods 145, 141-168. doi:10.1007/978-1-4939-9228-7_8
Nimtz, L., Hartmann, J., Tigges, J. et al. (2020). Characterization and application of electrically active neuronal networks established from human induced pluripotent stem cell-derived neural progenitor cells for neurotoxicity evaluation. Stem Cell Res 45, 101761. doi:10.1016/j.scr.2020.101761
NRC – National Research Council (2000). Scientific Frontiers in Developmental Toxicology and Risk Assessment. National Academies Press. doi:10.17226/9871
Nyffeler, J., Dolde, X., Krebs, A. et al. (2017). Combination of multiple neural crest migration assays to identify environmental toxicants from a proof-of-concept chemical library. Arch Toxicol 91, 3613-3632. doi:10.1007/s00204-017-1977-y
Ockleford, C., Adriaanse, P., Hougaard Bennekou, S. et al. (2018). Scientific opinion on pesticides in foods for infants and young children. EFSA J 16, e05286. doi:10.2903/j.efsa.2018.5286
Odawara, A., Katoh, H., Matsuda, N. et al. (2016). Physiological maturation and drug responses of human induced pluripotent stem cell-derived cortical neuronal networks in long-term culture. Sci Rep 6, 26181. doi:10.1038/srep26181
Odawara, A., Matsuda, N., Ishibashi, Y. et al. (2018). Toxicological evaluation of convulsant and anticonvulsant drugs in human induced pluripotent stem cell-derived cortical neuronal networks using an MEA system. Sci Rep 8, 10416. doi:10.1038/s41598-018-28835-7
OECD (2007). Test No. 426: Developmental Neurotoxicity Study. OECD Guidelines for the Testing of Chemicals, Section 4: Health Effects. http://www.oecd.org/dataoecd/20/52/37622194.pdf (accessed 22.07.2021)
Okado, N., Kakimi, S. and Kojima, T. (1979). Synaptogenesis in the cervical cord of the human embryo: Sequence of synapse formation in a spinal reflex pathway. J Comp Neurol 184, 491-517. doi:10.1002/cne.901840305
Oulhote, Y. and Bouchard, M. F. (2013). Urinary metabolites of organophosphate and pyrethroid pesticides and behavioral problems in Canadian children. Environ Health Perspect 121, 1378-1384. doi:10.1289/ehp.1306667
Paolicelli, R. C. and Ferretti, M. T. (2017). Function and dysfunction of microglia during brain development: Consequences for synapses and neural circuits. Front Synaptic Neurosci 9, 9. doi:10.3389/fnsyn.2017.00009
Paparella, M., Bennekou, S. H. and Bal-Price, A. (2020). An analysis of the limitations and uncertainties of in vivo developmental neurotoxicity testing and assessment to identify the potential for alternative approaches. Reprod Toxicol 96, 327-336. doi:10.1016/j.reprotox.2020.08.002
Pistollato, F., Carpi, D., Mendoza-de Gyves, E. et al. (2021). Combining in vitro assays and mathematical modelling to study developmental neurotoxicity induced by chemical mixtures. Reprod Toxicol 105, 101-119. doi:10.1016/j.reprotox.2021.08.007
Pitzer, E. M., Williams, M. T. and Vorhees, C. V. (2021). Effects of pyrethroids on brain development and behavior: Deltamethrin. Neurotoxicol Teratol 87, 106983. doi:10.1016/j.ntt.2021.106983
Radio, N. M., Breier, J. M., Shafer, T. J. et al. (2008). Assessment of chemical effects on neurite outgrowth in PC12 cells using high content screening. Toxicol Sci 105, 106-118. doi:10.1093/toxsci/kfn114
Robinette, B. L., Harrill, J. A., Mundy, W. R. et al. (2011). In vitro assessment of developmental neurotoxicity: Use of microelectrode arrays to measure functional changes in neuronal network ontogeny. Front Neuroeng 4, 1-9. doi:10.3389/fneng.2011.00001
Rodier, P. M. (1995). Developing brain as a target of toxicity. Environ Health Perspect 103, Suppl 6, 73-76. doi:10.1289/ehp.95103s673
Saavedra, L., Wallace, K., Freudenrich, T. F. et al. (2021). Comparison of acute effects of neurotoxic compounds on network activity in human and rodent neural cultures. Toxicol Sci 180, 295-312. doi:10.1093/toxsci/kfab008
Sachana, M., Bal-Price, A., Crofton, K. M. et al. (2019). International regulatory and scientific effort for improved developmental neurotoxicity testing. Toxicol Sci 167, 45-57. doi:10.1093/toxsci/kfy211
Sagiv, S. K., Thurston, S. W., Bellinger, D. C. et al. (2010). Prenatal organochlorine exposure and behaviors associated with attention deficit hyperactivity disorder in school-aged children. Am J Epidemiol 171, 593-601. doi:10.1093/aje/kwp427
Schmuck, M. R., Temme, T., Dach, K. et al. (2017). Omnisphero: A high-content image analysis (HCA) approach for phenotypic developmental neurotoxicity (DNT) screenings of organoid neurosphere cultures in vitro. Arch Toxicol 91, 2017-2028. doi:10.1007/s00204-016-1852-2
Seki, T. (2002). Expression patterns of immature neuronal markers PSA-NCAM, CRMP-4 and NeuroD in the hippocampus of young adult and aged rodents. J Neurosci Res 70, 327-334. doi:10.1002/jnr.10387
Shafer, T. J., Rijal, S. O. and Gross, G. W. (2008). Complete inhibition of spontaneous activity in neuronal networks in vitro by deltamethrin and permethrin. Neurotoxicology 29, 203-212. doi:10.1016/j.neuro.2008.01.002
Shafer, T. J. (2019). Application of microelectrode array approaches to neurotoxicity testing and screening. Adv Neurobiol 22, 275-297. doi:10.1007/978-3-030-11135-9_12
Sheets, L. P., Li, A. A., Minnema, D. J. et al. (2016). A critical review of neonicotinoid insecticides for developmental neurotoxicity. Crit Rev Toxicol 46, 153-190. doi:10.3109/10408444.2015.1090948
Shelton, J. F., Geraghty, E. M., Tancredi, D. J. et al. (2014). Neurodevelopmental disorders and prenatal residential proximity to agricultural pesticides: The charge study. Environ Health Perspect 122, 1103-1109. doi:10.1289/ehp.1307044
Smirnova, L., Hogberg, H. T., Leist, M. et al. (2014). Food for thought ... Developmental neurotoxicity – Challenges in the 21st century and in vitro opportunities. ALTEX 31, 129-156. doi:10.14573/altex.1403271
Stern, M., Gierse, A., Tan, S. et al. (2014). Human Ntera2 cells as a predictive in vitro test system for developmental neurotoxicity. Arch Toxicol 88, 127-136. doi:10.1007/S00204-013-1098-1
Takahashi, K., Tanabe, K., Ohnuki, M. et al. (2007). Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 131, 861-872. doi:10.1016/j.cell.2007.11.019
Tapia, C. M., Folorunso, O., Singh, A. K. et al. (2020). Effects of deltamethrin acute exposure on Nav1.6 channels and medium spiny neurons of the nucleus accumbens. Toxicology 440, 152488. doi:10.1016/j.tox.2020.152488
Terron, A. and Bennekou, S. H. (2018). Towards a regulatory use of alternative developmental neurotoxicity testing (DNT). Toxicol Appl Pharmacol 354, 19-23. doi:10.1016/j.taap.2018.02.002
Toscano, C. D. and Guilarte, T. R. (2005). Lead neurotoxicity: From exposure to molecular effects. Brain Res Rev 49, 529-554. doi:10.1016/j.brainresrev.2005.02.004
Tsuji, R. and Crofton, K. M. (2012). Developmental neurotoxicity guideline study: Issues with methodology, evaluation and regulation*. Congenit Anom (Kyoto) 52, 122-128. doi:10.1111/J.1741-4520.2012.00374.X
Tukker, A. M., Bouwman, L. M. S., van Kleef, R. G. D. M. et al. (2020a). Perfluorooctane sulfonate (PFOS) and perfluorooctanoate (PFOA) acutely affect human α1β2γ2L GABAA receptor and spontaneous neuronal network function in vitro. Sci Rep 10, 5311. doi:10.1038/s41598-020-62152-2
Tukker, A. M., Wijnolts, F. M. J., de Groot, A. et al. (2020b). Applicability of hiPSC-derived neuronal co-cultures and rodent primary cortical cultures for in vitro seizure liability assessment. Toxicol Sci 178, 71-87. doi:10.1093/toxsci/kfaa136
Uhlhaas, P. and Singer, W. (2006). Neural synchrony in brain disorders: Relevance for cognitive dysfunctions and pathophysiology. Neuron 52, 155-168. doi:10.1016/j.neuron.2006.09.020
U.S. EPA (1998). Health Effects Guidelines OPPTS 870.6300. Dev Neurotox Study EPA 71.
Wagner-Schuman, M., Richardson, J. R., Auinger, P. et al. (2015). Association of pyrethroid pesticide exposure with attention-deficit/hyperactivity disorder in a nationally representative sample of U.S. children. Environ Health 14, 44. doi:10.1186/s12940-015-0030-y
Xue, Z., Li, X., Su, Q. et al. (2013). Effect of synthetic pyrethroid pesticide exposure during pregnancy on the growth and development of infants. Asia Pac J Public Health 25, Suppl 4, 72S-79S. doi:10.1177/1010539513496267
Zhang, L. I. and Poo, M. M. (2001). Electrical activity and development of neural circuits. Nat Neurosci 4, Suppl, 1207-1214. doi:10.1038/NN753
Zhang, R., Zhang, H., Chen, B. et al. (2020). Fetal bovine serum attenuating perfluorooctanoic acid-inducing toxicity to multiple human cell lines via albumin binding. J Hazard Mater 389, 122109. doi:10.1016/j.jhazmat.2020.122109
Zhang, Y., Xu, Y.-Y., Sun, W.-J. et al. (2016). FBS or BSA inhibits EGCG induced cell death through covalent binding and the reduction of intracellular ROS production. Biomed Res Int 2016, 5013409. doi:10.1155/2016/5013409